What Affects Image Quality in Modern Ophthalmic Equipment?
Image quality in modern ophthalmic equipment is shaped by a chain of interacting technical variables, not by pixel count alone.
Across healthcare technology and broader B2B evaluation contexts, imaging performance affects diagnosis confidence, device usability, upgrade cycles, and long-term service value.
When comparing ophthalmic equipment, it is essential to assess optics, sensors, illumination, processing, calibration, and workflow integration as one connected system.
A device may advertise high resolution, yet still produce weak clinical images if contrast, stability, or repeatability are compromised.
This article explains what affects image quality in modern ophthalmic equipment and how to evaluate those factors in practical, cross-functional terms.
Core Factors Behind Image Quality in Ophthalmic Equipment
The first layer of image quality in ophthalmic equipment begins with optical capture and signal integrity.
Every step, from incoming light to displayed image, can add detail or remove it.
Optical design and lens quality
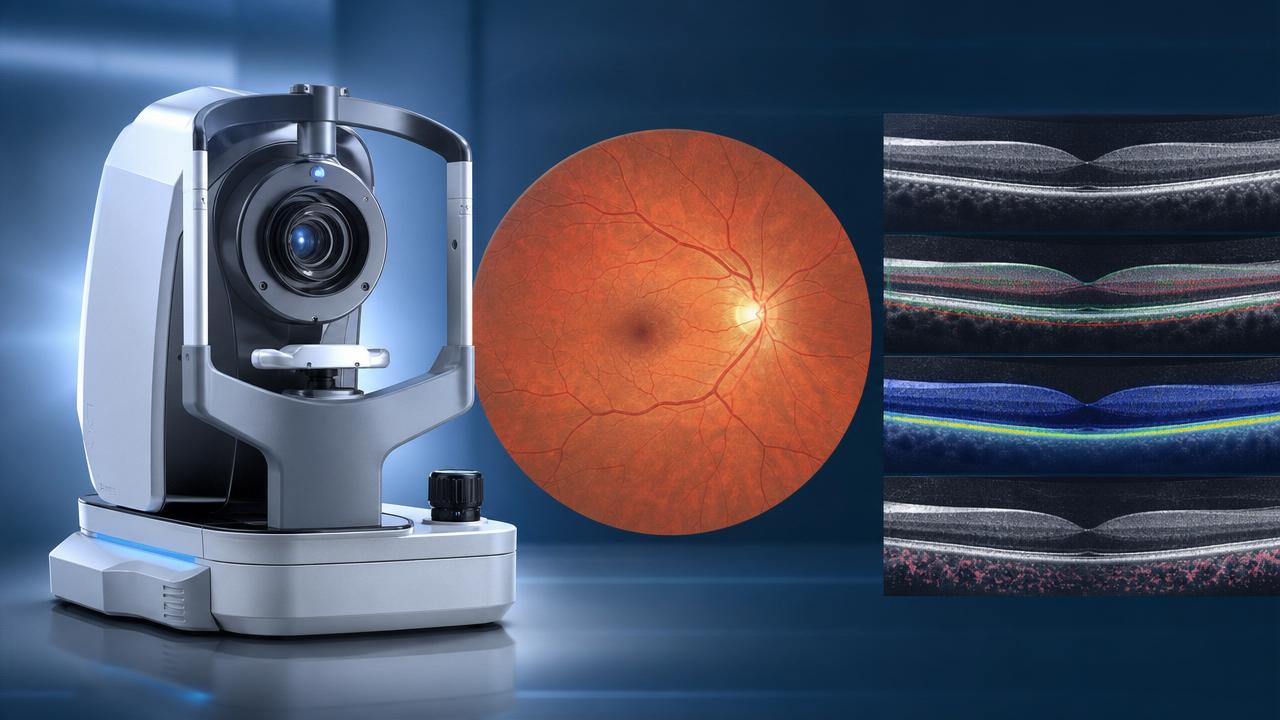
Well-designed optics determine sharpness, field uniformity, distortion control, and color fidelity.
In ophthalmic equipment, poor lens coatings or alignment can create flare, chromatic aberration, and edge softness.
These issues reduce visibility of retinal structures, corneal boundaries, or subtle pathological patterns.
Sensor performance and signal-to-noise ratio
Sensor quality strongly influences sensitivity, dynamic range, and low-light performance.
A larger or more efficient sensor can preserve fine detail while reducing noise in dim or fast-capture conditions.
In ophthalmic equipment, cleaner signals support better visualization of microstructures and vessel contrast.
Illumination stability
Light source consistency affects exposure control, color repeatability, and artifact reduction.
Unstable illumination can introduce flicker, hotspots, or uneven brightness across the image.
For ophthalmic equipment, this matters when comparing baseline images over time or across sites.
Image processing algorithms
Modern ophthalmic equipment often relies on software to sharpen, denoise, segment, and enhance images.
Effective algorithms improve interpretability without masking clinically meaningful features.
Overaggressive processing may create artificial edges, suppress subtle findings, or distort tissue appearance.
Calibration and mechanical precision
Even advanced ophthalmic equipment can underperform if calibration drifts or moving components lose precision.
Focus accuracy, scan alignment, positioning stability, and optical centering directly affect repeatable image quality.
Why the Industry Focuses on More Than Resolution
The healthcare technology market increasingly evaluates ophthalmic equipment through total imaging reliability rather than headline specifications.
This shift reflects tighter quality expectations, digital workflow demands, and broader data comparability needs.
- Higher image volumes require stable performance across operators and locations.
- Longitudinal care depends on repeatable brightness, scale, and anatomical consistency.
- AI-assisted analysis needs clean, standardized image input.
- Serviceability and recalibration now influence total lifecycle value.
In this environment, ophthalmic equipment is assessed as a data-producing asset, not only as a viewing device.
That perspective is also relevant in comprehensive industry analysis, where equipment quality affects interoperability, procurement efficiency, and future platform compatibility.
Business and Operational Value of Better Image Quality
High-performing ophthalmic equipment creates value beyond the image itself.
Better image quality reduces retakes, shortens examination time, and supports more dependable reporting.
It also strengthens confidence when devices are used across networked healthcare settings or multi-site service models.
Diagnostic confidence
Reliable ophthalmic equipment helps preserve clinically relevant details without visual ambiguity.
This is especially important when minor changes influence follow-up decisions or referral pathways.
Workflow efficiency
Fewer failed captures mean less operator adjustment and smoother throughput.
In ophthalmic equipment, autofocus speed, alignment aids, and exposure automation can directly affect usable image yield.
Lifecycle economics
Devices with stable image quality often require fewer corrective interventions and support longer productive use.
That makes ophthalmic equipment evaluation relevant to maintenance planning, software updates, and training strategy.
Typical Imaging Scenarios and Quality Priorities
Not all ophthalmic equipment is judged by the same imaging priorities.
Different applications emphasize different trade-offs between speed, depth, contrast, field of view, and repeatability.
This comparison shows why ophthalmic equipment should be matched to intended use, not selected by specifications in isolation.
Practical Assessment Points for Comparing Ophthalmic Equipment
A structured review process helps translate technical claims into usable evaluation criteria.
The following checkpoints are useful when comparing ophthalmic equipment across product lines or deployment scenarios.
- Request sample images from multiple conditions, not only ideal demonstrations.
- Check edge-to-edge sharpness and uniform exposure.
- Review performance under low signal or mild motion.
- Confirm whether image enhancement is reversible or traceable.
- Assess recalibration frequency and service documentation.
- Verify export quality for EMR, PACS, and analytics platforms.
- Examine how consistently the ophthalmic equipment performs across users.
These points help separate attractive specifications from dependable real-world imaging performance.
Implementation Considerations and Next-Step Review
Image quality in ophthalmic equipment should be reviewed as part of a broader operational and digital strategy.
The best choice usually combines optical integrity, robust software, calibration discipline, and practical interoperability.
It is useful to document required image outputs, expected volume, service expectations, and integration needs before final comparison.
For organizations tracking healthcare technology trends through platforms such as TradeNexus Pro, this approach supports clearer benchmarking and more durable equipment decisions.
When reviewing ophthalmic equipment, focus on repeatable image quality, not isolated technical claims, and align each feature with measurable diagnostic and workflow outcomes.
Get weekly intelligence in your inbox.
No noise. No sponsored content. Pure intelligence.