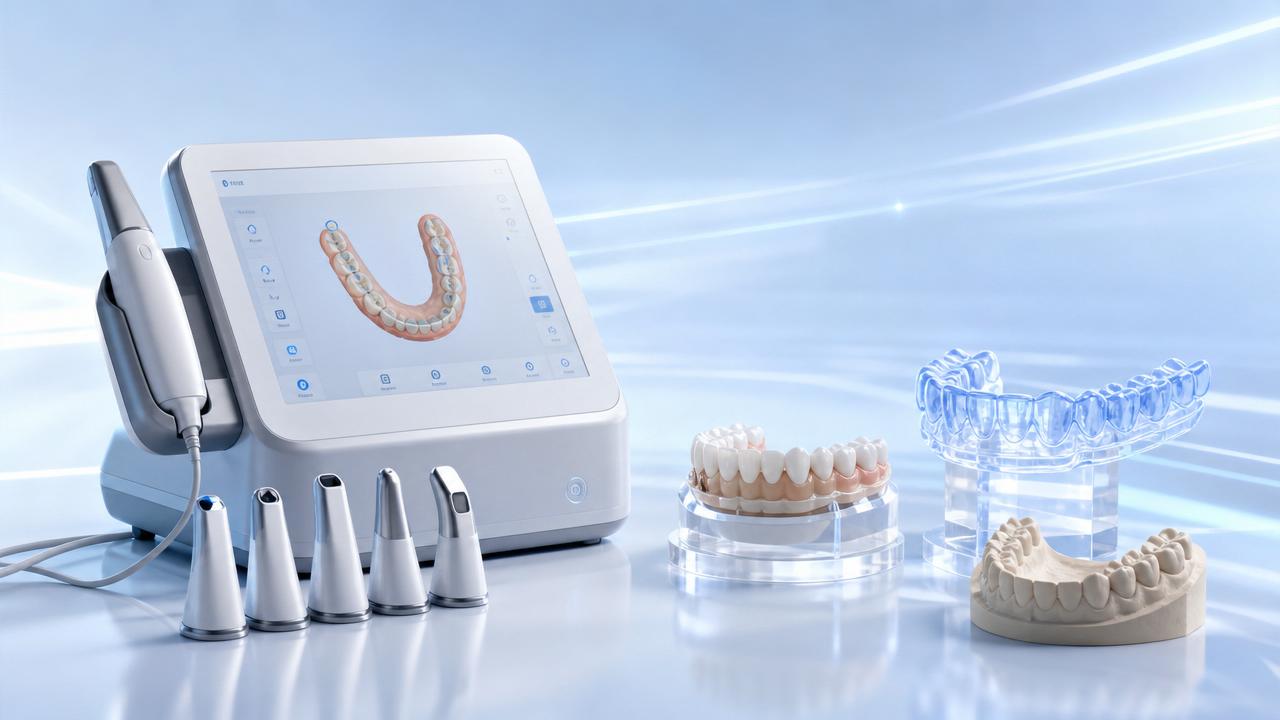
Clear Aligner Manufacturing: Why Consistency Is Hard to Scale
In clear aligner manufacturing, scale does not fail because demand is too high; it fails when repeatability breaks at each hidden handoff. A workflow may look digital from scan to shipment, yet small shifts in sheet thickness, printer calibration, trimming tolerance, or batch tracking can compound into fit deviations that are costly to catch late. For teams building output across multiple lines, sites, or supplier networks, the central question is not simply how to produce more trays, but how to preserve process discipline while throughput rises. That is why clear aligner manufacturing is best understood through scenarios: pilot operations, rapid-growth facilities, multi-site production, and regulated export environments each expose different consistency risks and require different control strategies.
When clear aligner manufacturing moves from pilot runs to scale-up, variability changes shape

Early-stage clear aligner manufacturing often appears stable because output volumes are small, operators are highly attentive, and exception handling is still manual. In that scenario, technicians can compensate for print defects, thermoforming drift, or packaging errors before they become systemic. Once volume expands, the same operation faces a different reality: more shifts, more equipment utilization, more incoming material lots, and more digital files moving through automated queues. What looked like isolated defects becomes a statistical pattern.
This is why consistency is hard to scale in clear aligner manufacturing. The difficulty is not only technical; it is procedural. A highly accurate CAD setup can still produce inconsistent aligners if support design changes are not version-controlled, if model orientation differs across operators, or if post-processing exposure times vary between machines. At higher output, process discipline becomes as important as equipment capability.
Scenario 1: Prototype-heavy clear aligner manufacturing needs flexibility, not yet maximum automation
In low-volume or development-focused clear aligner manufacturing, the main objective is fast iteration. Materials may still be under evaluation, treatment planning assumptions may be refined, and tooling strategies may change frequently. Here, flexibility matters more than cycle-time optimization. The core judgment point is whether variability comes from intentional experimentation or uncontrolled execution.
For this scenario, the most useful controls are digital traceability and baseline validation. Every aligner stage should be linked to the source file version, print profile, resin batch, thermoforming settings, and finishing route. If a fit issue appears, the team should be able to identify whether the root cause came from design logic, printed model accuracy, material response, or manual trimming. In clear aligner manufacturing, this early discipline prevents bad habits from scaling into expensive process instability later.
Core judgment points
- Are process changes documented at every revision?
- Is model dimensional accuracy measured against a fixed benchmark?
- Can defects be traced to one step rather than multiple uncertain variables?
Scenario 2: Fast-growth clear aligner manufacturing struggles most with handoff loss
The most common scale-up problem in clear aligner manufacturing appears when order growth outruns process standardization. A facility adds printers, increases thermoforming stations, and hires more operators, but the workflow architecture remains fragmented. Design files may arrive in different naming formats, print queues may not prioritize by due date and complexity, and quality criteria may depend too heavily on individual judgment. The result is not one catastrophic failure, but a steady rise in rework, remakes, and cycle-time unpredictability.
In this scenario, the core judgment point is where process knowledge resides. If consistency depends on a few experienced technicians, then clear aligner manufacturing is not yet scalable. Robust scale requires codified SOPs, locked machine recipes, barcode-based routing, and in-line verification checkpoints. Thermoforming pressure, heating time, cooling behavior, and trimming references should not vary by shift. Even packaging and labeling need verification, because patient-specific products carry zero tolerance for identity mismatch.
A practical upgrade path is to map every handoff from digital treatment plan to final dispatch and identify where information is re-entered manually. Each manual re-entry introduces risk. Reducing those touchpoints often delivers better consistency in clear aligner manufacturing than adding another machine.
Scenario 3: Multi-site clear aligner manufacturing requires identical outputs from non-identical environments
When production expands across more than one site, clear aligner manufacturing becomes a network problem. Equipment may be the same on paper, yet environmental conditions, operator training, maintenance discipline, and supplier lead times can differ. One site may use the same thermoplastic brand but receive a different lot profile; another may maintain printers more frequently and produce more stable models. If sites are expected to deliver interchangeable output, process harmonization must be engineered rather than assumed.
The key judgment point in this scenario is whether equivalence has been proven. Shared SOPs alone are insufficient. Clear aligner manufacturing at multiple sites needs cross-site validation models, periodic gauge R&R studies, standardized acceptance thresholds, and a common data layer for deviation analysis. Without a centralized quality intelligence system, one site’s drift can remain hidden until customer complaints reveal it.
Signals that site-to-site consistency is at risk
- Different remake rates for similar case types
- Uneven dimensional results across printer fleets
- Inconsistent finishing quality after trimming or polishing
- Quality reviews based on local interpretation instead of shared metrics
Scenario 4: Regulated and export-facing clear aligner manufacturing raises the cost of inconsistency
For export programs or highly regulated channels, clear aligner manufacturing must balance throughput with auditable control. Here, inconsistency is not only an operational issue; it can become a compliance issue. Material traceability, device history records, packaging integrity, and labeling accuracy are part of product quality, not administrative overhead. The core judgment point is whether the quality system can withstand external review under production pressure.
This scenario requires more than final inspection. Stable clear aligner manufacturing depends on preventive controls: validated equipment settings, documented change control, nonconformance workflows, and controlled supplier qualification. If a resin, film, or trimming tool changes, the downstream effect on fit and durability must be assessed before the change is released broadly. The larger the market footprint, the more expensive uncontrolled variability becomes.
How clear aligner manufacturing needs differ by production scenario
Scenario-fit recommendations for more stable clear aligner manufacturing
- For pilot operations: establish a golden build with fixed file preparation, print orientation, curing settings, and thermoforming parameters before expanding SKU volume.
- For growth-stage lines: prioritize MES-style tracking, barcode routing, and machine recipe locking before purchasing more capacity.
- For multi-site systems: run the same validation case set across all facilities and compare dimensional, finishing, and remake outcomes monthly.
- For regulated production: align engineering change control with supplier qualification so material substitutions never bypass validation.
- For any clear aligner manufacturing environment: monitor leading indicators, not only final defects, including print drift, forming scrap, trim adjustment frequency, and queue interruptions.
Common misjudgments that weaken clear aligner manufacturing consistency
One frequent mistake is assuming that higher equipment precision automatically guarantees stable clear aligner manufacturing. In reality, premium printers and forming systems still depend on material control, preventive maintenance, and repeatable digital preparation. Another misjudgment is overreliance on final inspection. By the time a fit problem is visible in the finished aligner, the true cause may be several steps upstream and already affecting many units.
A third blind spot is underestimating data fragmentation. If print logs, QC records, and shipment data live in separate systems, process learning slows down. Clear aligner manufacturing improves fastest when quality data is connected end to end. The final error is treating remakes as a normal cost of growth. In a high-volume environment, remakes are not just waste; they are evidence that the process is teaching the wrong lesson unless the root cause is closed systematically.
What to do next if clear aligner manufacturing consistency is the real bottleneck
Start with a scenario-based audit instead of a generic capacity review. Map where clear aligner manufacturing variability is most likely to emerge in the current operating model: digital design preparation, model printing, thermoforming, trimming, packaging, or cross-site transfer. Then rank each step by impact on fit, traceability, and remake risk. This approach reveals whether the next investment should go into automation, validation, software integration, training control, or supplier management.
For organizations following advanced manufacturing and healthcare technology supply chains through platforms such as TradeNexus Pro, the strongest signal is not who can produce more aligners today, but who can scale clear aligner manufacturing with documented repeatability tomorrow. Consistency is the capability that converts output into trust. When that capability is built scenario by scenario, scale becomes much easier to sustain.
Get weekly intelligence in your inbox.
No noise. No sponsored content. Pure intelligence.