Laboratory Incubators: When Temperature Uniformity Becomes a Problem
Laboratory incubators are built to maintain stable environmental conditions, yet temperature uniformity often becomes the hidden variable that undermines performance. When one part of the chamber runs warmer or cooler than another, cell cultures, microbiology samples, reagents, and test workflows can all be affected in ways that are difficult to detect until results begin to drift.
For information-focused readers evaluating laboratory incubators, the key point is simple: poor temperature uniformity is not a minor specification issue. It directly influences sample integrity, repeatability, qualification outcomes, and the long-term cost of ownership. Understanding what causes non-uniformity, how to verify it, and when it signals a design or application mismatch is essential for both lab users and technical buyers.
Why Temperature Uniformity Matters More Than Many Buyers Expect
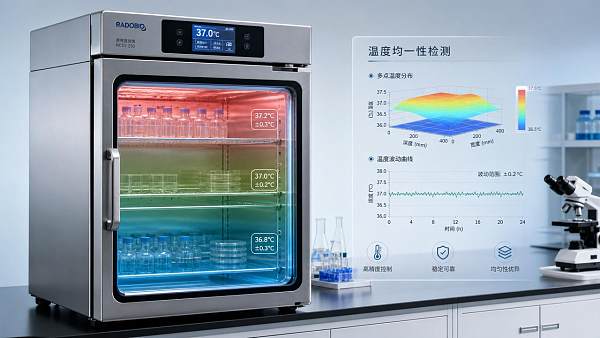
Most buyers first look at setpoint range, chamber volume, and basic accuracy. Those specifications matter, but temperature uniformity is often more revealing because it reflects how evenly the incubator performs across the usable space, not just what temperature appears on the display.
If an incubator is set to 37°C, that does not automatically mean every shelf position is truly operating at 37°C. In practice, some areas may run hotter near heaters or fans, while corners, door zones, or heavily loaded sections may run cooler. Even a modest variation can create meaningful differences in growth conditions or test outcomes.
For microbiology, tissue culture, stability work, and sensitive analytical preparation, these deviations can reduce confidence in experimental consistency. A process may appear stable when measured at a single point, yet still expose samples to uneven conditions over time. That gap between displayed temperature and actual chamber-wide performance is where many problems begin.
From a procurement perspective, this means a low-cost unit with weak uniformity may create higher downstream costs through failed batches, repeated tests, calibration issues, or user complaints. In other words, the business impact of poor uniformity often exceeds the original equipment price difference.
What Searchers Usually Want to Know When Uniformity Becomes a Problem
Readers searching this topic are rarely looking for a textbook definition alone. Their underlying search intent is usually diagnostic and practical. They want to understand why a laboratory incubator that should be stable is delivering inconsistent conditions, whether the issue is serious, and what actions should be taken next.
In most cases, target readers care about five questions. First, how much non-uniformity is acceptable for their application? Second, what are the most likely root causes? Third, can the issue be corrected through operation, loading, maintenance, or calibration? Fourth, how should equipment be evaluated before purchase? Fifth, when does the problem indicate that a different incubator design is needed?
That is why useful content on laboratory incubators should not spend too much time on generic product overviews. Readers benefit more from guidance that helps them interpret risk, compare equipment meaningfully, and decide whether the problem is procedural, mechanical, or structural to the unit itself.
What Causes Temperature Uniformity Problems in Laboratory Incubators?
Temperature non-uniformity usually comes from a combination of design limitations, environmental influences, and user practices. Rarely is there a single cause in isolation. Understanding the interaction between these factors is the fastest way to reach a sound conclusion.
Airflow design is one of the most important variables. In forced-convection incubators, fans help distribute heat, but fan placement, ducting, and circulation pattern determine whether the chamber temperature actually becomes even. Poor airflow architecture can create hot spots, cold spots, and recovery differences across shelf levels.
Heater location and control logic also matter. If heat is introduced unevenly or the controller reacts slowly, the incubator may overshoot in one area while lagging in another. Some units hold a stable average temperature but still perform unevenly spatially, especially during door openings or load changes.
Door design and sealing are frequent contributors. Worn gaskets, repeated access, or weak inner door sealing can allow localized heat loss. This is especially important in busy labs where doors are opened often. The temperature display may recover quickly, but actual sample-level recovery can be much slower.
Shelf loading and sample placement are often underestimated. Overloading shelves, blocking vents, clustering dense materials, or placing large water-containing loads in one area can disrupt circulation. In these situations, the incubator itself may not be defective; the load pattern is changing how heat moves through the chamber.
Ambient room conditions can also influence performance. Units installed near HVAC vents, direct sunlight, exterior walls, autoclaves, or other heat-generating equipment may experience external thermal stress. Even a well-designed incubator can struggle to maintain chamber-wide consistency in a poor installation environment.
Maintenance and aging should not be ignored. Fan wear, sensor drift, damaged seals, dust accumulation, and neglected calibration all contribute to performance deterioration. An incubator that originally met uniformity expectations may no longer do so after years of use.
How Uniformity Problems Affect Sample Integrity and Lab Performance
The practical consequences depend on the application, but they are often more serious than users initially assume. In biological workflows, temperature gradients may alter growth rates, viability, metabolism, or contamination dynamics. In microbiology, this can lead to misleading colony behavior or inconsistent incubation endpoints.
In quality-sensitive environments, the risk expands beyond the sample itself. Temperature variation can compromise method repeatability, create batch-to-batch inconsistency, and trigger investigation cycles that consume both labor and materials. If the incubator supports validation-related work, poor uniformity may create documentation issues and qualification delays.
Another challenge is that non-uniformity can mimic other problems. Teams may suspect media quality, operator technique, or reagent variability before recognizing that sample placement inside the chamber is the real driver. This delays corrective action and increases the cost of troubleshooting.
For buyers and lab managers, the broader lesson is clear: temperature uniformity is not only a technical metric. It is a reliability metric, a quality metric, and in many facilities, a cost-control metric.
How to Tell Whether the Problem Is Real or Just a Measurement Artifact
Before concluding that an incubator is underperforming, users should confirm that the apparent problem is being measured correctly. A display reading alone is not sufficient. Chamber mapping with multiple calibrated probes is usually the best way to understand actual spatial variation.
Measurements should be taken at different locations, including corners, center positions, upper and lower shelves, and areas near the door. It is also important to test under realistic operating conditions. An empty chamber may look more uniform than a chamber loaded with actual containers, media, or product.
Timing matters as well. Readings taken immediately after door opening, startup, or setpoint adjustment can exaggerate temporary variation. A proper evaluation should include stabilized conditions and, when relevant, recovery performance after disturbance.
Users should also distinguish between accuracy, stability, and uniformity. An incubator may be accurate at the control sensor, stable over time at one point, yet still non-uniform across the chamber. These are related but different performance characteristics, and confusing them often leads to poor buying decisions.
When Can the Issue Be Fixed Operationally?
Not every uniformity problem requires replacement. In many labs, performance improves significantly when operating practices are corrected. The most common improvement area is load management. Shelves should not block circulation paths, and large or thermally dense items should be distributed evenly rather than concentrated in one zone.
Door discipline can also make a measurable difference. Frequent or prolonged opening introduces repeated disturbances that some incubators cannot recover from evenly. If workflows require constant access, the issue may be procedural rather than purely mechanical.
Routine maintenance is another practical lever. Replacing gaskets, cleaning airflow paths, checking fan function, and verifying sensor calibration can restore expected behavior. For critical applications, periodic mapping should be part of preventive quality practice rather than a one-time response to problems.
That said, operational fixes have limits. If repeated mapping shows persistent gradients despite proper loading, stable room conditions, and completed maintenance, the root problem may be inherent to the unit design or to an application mismatch.
What Buyers Should Ask Before Choosing Laboratory Incubators
For technical buyers, the most useful evaluation approach is to move beyond headline specifications and ask performance questions tied to real use. A supplier should be able to explain how temperature uniformity was tested, under what load conditions, at what ambient environment, and across which chamber points.
It is also worth asking whether published values reflect an empty chamber or a loaded chamber. Empty-chamber data can look impressive but may not represent daily use. Buyers should request recovery data after door opening, not just static equilibrium numbers.
Other useful questions include: How is airflow managed? Where are heaters and sensors positioned? What maintenance intervals are recommended? How does chamber size affect uniformity? What application types is the model best suited for? Can the vendor provide mapping support, validation documentation, or service responsiveness in the target region?
For organizations comparing multiple laboratory incubators, a simple scorecard can help. Rate each option on uniformity specification quality, test transparency, service support, application fit, energy efficiency, and total lifecycle risk. This makes selection less vulnerable to marketing claims and more aligned with operational value.
When Poor Uniformity Means You Need a Different Incubator Type
Sometimes the problem is not that the incubator is malfunctioning. It is that the chosen model is unsuitable for the workload. A basic incubator may be acceptable for routine tasks but insufficient for sensitive culture work, high-volume throughput, or regulated testing environments.
For example, applications with strict consistency demands may require a higher-grade forced-convection design, better chamber insulation, tighter control systems, or specialized features for contamination control and recovery performance. Large chambers may also require more careful design to maintain even conditions throughout the usable space.
If a lab routinely handles variable loads, frequent access, or temperature-sensitive samples positioned across multiple shelves, the purchase decision should prioritize real-world uniformity over nominal capacity or low upfront price. In many cases, investing in the right design early prevents recurring operational headaches later.
A Practical Decision Framework for Researchers and Procurement Teams
If temperature uniformity becomes a concern, the most effective response is structured and evidence-based. First, verify the issue with multi-point measurement. Second, review loading patterns, door-opening behavior, and room conditions. Third, complete maintenance and calibration checks. Fourth, compare measured performance against actual application requirements rather than generic expectations.
If the incubator still fails to support the intended process, the decision becomes strategic. The lab must determine whether the equipment can be upgraded, requalified for a less demanding use, or replaced with a model better matched to technical and business requirements.
For information researchers and B2B buyers, this is where credible market intelligence matters. Product selection should not rely only on brochure claims. It should combine technical specifications, application fit, service capability, and risk awareness. The most valuable purchase is not the incubator with the longest feature list, but the one that delivers dependable thermal consistency in the real operating environment.
Conclusion
When temperature uniformity becomes a problem in laboratory incubators, the issue should be treated as a meaningful performance risk, not a minor inconvenience. Uneven chamber conditions can affect sample quality, repeatability, validation confidence, and total operating cost.
The best response is to understand the source of the variation, verify it with proper measurement, and separate fixable operational factors from deeper equipment limitations. For both researchers and procurement teams, better decisions come from asking sharper questions about actual chamber-wide performance.
In the end, laboratory incubators should be judged not just by the temperature they display, but by the consistency they deliver where samples truly sit. That is the standard that protects data integrity, supports reliable workflows, and justifies long-term investment.
Get weekly intelligence in your inbox.
No noise. No sponsored content. Pure intelligence.