Dental Implant Kits: What Often Gets Missed Before Ordering
Ordering dental implant kits may seem straightforward, but procurement teams often overlook critical factors that affect clinical compatibility, regulatory compliance, supplier reliability, and long-term cost control. For buyers aiming to reduce risk and improve sourcing outcomes, understanding what gets missed before purchasing dental implant kits is essential to making smarter, more strategic decisions.
In B2B healthcare purchasing, the challenge is rarely limited to unit price. A dental implant kit can influence procedure workflow, inventory planning, sterilization protocol, clinician adoption, and after-sales support over a 12- to 36-month sourcing cycle. For procurement professionals evaluating dental implant kits across distributors, OEM suppliers, or cross-border vendors, the real value lies in what is verified before the purchase order is issued.
This article outlines the practical details that often get missed, from component completeness and traceability to packaging integrity, training needs, and hidden lifecycle costs. The goal is to help buyers reduce rework, avoid mismatched systems, and build a more resilient sourcing process for dental implant kits.
Why Dental Implant Kit Procurement Is More Complex Than It Looks
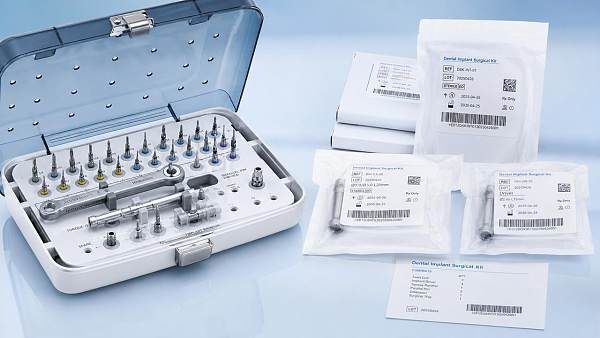
A dental implant kit is not a single product in the usual sense. In many sourcing situations, it is a bundled procedural system that may include drills, drivers, torque tools, depth indicators, impression components, surgical guides, and sterilizable trays. Missing even 1 critical component can delay treatment schedules, create compatibility issues, or force emergency local purchasing at a significantly higher cost.
For procurement teams, the most common mistake is buying by headline description rather than by bill-of-components verification. Two suppliers may both list “dental implant kits,” yet one may include 18 reusable surgical items and 4 prosthetic interfaces, while another includes only the core drilling sequence and excludes torque instruments or replacement parts. That gap directly affects total acquisition cost.
The difference between catalog similarity and clinical usability
On paper, many dental implant kits appear functionally equivalent. In practice, usability depends on drill sequence logic, implant platform matching, color coding consistency, and whether the kit aligns with the clinician’s preferred protocol. If the surgical sequence requires 5 drilling steps but the supplied kit supports only 4, workflow disruption becomes immediate.
Another overlooked factor is whether the kit is intended for starter cases, full restorative workflows, or guided surgery. Buyers should distinguish between at least 3 common categories: surgical-only kits, restorative kits, and integrated systems. Mixing these categories during RFQ comparison often leads to inaccurate cost benchmarking.
What procurement should verify before comparing quotes
Before evaluating price, buyers should standardize the comparison framework. A useful first-pass checklist includes:
- Component count and exact instrument list
- Compatibility with implant platform, diameter range, and connection type
- Reusable versus single-use items
- Sterilization method and cycle tolerance
- Availability of replacement drills and consumables within 7–30 days
- Documentation for traceability, labeling, and market access
Without these 6 checks, procurement comparisons tend to reward incomplete offers that look cheaper at first but generate higher downstream costs in months 3 to 12.
The Most Frequently Missed Factors Before Ordering
The biggest sourcing errors usually occur in the pre-order stage. Buyers focus on price tiers, MOQ, and lead time, but skip technical validation. For dental implant kits, that is risky because product performance depends on system compatibility rather than on one standalone item.
1. Platform and connection compatibility
A common problem is assuming “universal” compatibility. Many dental implant kits are designed around specific implant diameters, hex structures, conical connections, or proprietary driver interfaces. If a clinic network uses 2 implant platforms across different locations, the procurement team must confirm whether one kit supports both or whether separate SKUs are required.
Ask for compatibility documentation in writing. A supplier should be able to clarify drill sequence range, supported implant sizes, and whether restorative components are cross-compatible or platform-specific. If that information is vague, post-purchase returns can become difficult or impossible.
Practical checkpoint
Require a validated item matrix showing platform type, diameter range, and interface match. Even a 0.2 mm mismatch in tolerances or an incorrect driver geometry can compromise workflow efficiency and increase chairside troubleshooting.
2. Sterilization durability and instrument lifespan
Not every reusable component in dental implant kits performs equally over repeated sterilization cycles. Buyers often ask whether tools are autoclavable, but fail to ask how many cycles the drills, trays, or torque instruments can typically withstand before replacement is recommended. That omission can distort annual cost projections.
For multi-site procurement, replacement frequency matters. If one kit requires drill replacement after 20–30 uses while another performs reliably across a broader maintenance cycle, the cheaper upfront option may become more expensive over 4 quarters. Procurement teams should request care instructions, expected wear guidance, and replacement availability.
The table below highlights several pre-order risk areas that should be reviewed before committing to dental implant kits from a new or alternate supplier.
The key takeaway is that dental implant kits should be evaluated as managed systems, not as simple line items. A complete package with documented support often outperforms a low-price offer that lacks replaceable parts or clear traceability.
3. Regulatory fit across destination markets
Cross-border buyers must pay close attention to market-specific compliance and import documentation. The same dental implant kit may be acceptable in one region but require different labeling, registration support, or sterilization declarations in another. Procurement should verify not just certification status, but document completeness for the target country or distributor channel.
At minimum, confirm 4 documentation points: product labeling, lot traceability, instructions for use, and packaging integrity information. If the purchase involves private labeling or regional redistribution, the review should start before the first sample shipment, not after commercial stock is in transit.
4. Supplier continuity and spare-part logic
A surprising number of buyers validate factory capability but do not check continuity of replacement items. Dental implant kits are often purchased with a 6- to 12-month forecast, yet drills and drivers may need replenishment more frequently. If spare components require 45–60 days lead time while clinical demand runs weekly, service disruption becomes likely.
Ask suppliers whether they maintain spare-parts inventory, whether partial replenishment is available, and what their reorder threshold is. A vendor that can ship replacement accessories in 7–14 days offers very different operational security from one that only builds to order every 8 weeks.
How Procurement Teams Should Evaluate Dental Implant Kits
A stronger purchasing process starts with structured evaluation criteria. Rather than selecting dental implant kits based on one distributor proposal or a clinician preference alone, procurement teams should use a cross-functional review model involving purchasing, clinical leadership, quality management, and inventory operations.
A 5-step sourcing framework
- Define use case: surgical-only, restorative, guided, training, or mixed-site deployment.
- Map required components by procedure sequence and implant platform.
- Request technical files, sterilization guidance, and replacement policy.
- Run sample validation with clinicians and central sterilization staff.
- Score landed cost, lead time, continuity risk, and supplier responsiveness.
This 5-step framework helps procurement avoid “apples to oranges” comparisons. It also reduces the chance of discovering missing items only after kits have been distributed to treatment centers.
What should be included in the RFQ
An effective RFQ for dental implant kits should go beyond price and MOQ. Include instrument list, material information, sterilization instructions, recommended replacement intervals, packaging details, labeling samples, spare-part availability, and standard lead times. If possible, ask suppliers to separate one-time kit cost from recurring accessory cost over 12 months.
That split matters because total cost of ownership is often shaped by recurring items rather than by the tray itself. In many B2B healthcare categories, the difference between a manageable program and an inefficient one comes from replacement planning, not initial kit purchase price.
The following comparison model can help buyers score dental implant kits more consistently across multiple vendors.
Using a matrix like this creates a more defensible sourcing decision, especially when internal stakeholders are balancing clinical usability with procurement efficiency and quality assurance requirements.
Sample validation should not be skipped
For high-impact or multi-site purchases, a sample review period of 2–4 weeks is usually more valuable than a purely catalog-based decision. This allows clinicians to confirm sequence logic, sterilization teams to review tray handling, and procurement to verify packaging consistency against submitted documents.
Even if the trial is limited to 1 or 2 sites, it can reveal issues that would otherwise scale across 20 or more kits in a network rollout. These issues often include unclear engraving, awkward tray layout, missing replacement references, or driver mismatch during actual use.
Hidden Cost Drivers and Long-Term Procurement Risks
Price pressure is normal in procurement, but dental implant kits should be judged on total operational value. A low initial quote can conceal recurring accessory purchases, tool wear, delayed replenishment, and clinician adaptation time. These factors affect not just cost, but also procurement workload and service continuity.
Where total cost of ownership increases
There are at least 4 recurring cost drivers that buyers should model before supplier selection: replacement drills, breakage or wear-related accessory swaps, sterilization tray maintenance, and emergency local sourcing when the original vendor cannot replenish on time. In many cases, these costs emerge gradually over 6 to 12 months, which is why they are often missed at onboarding.
Training time is another hidden variable. If one dental implant kit uses a non-intuitive layout or inconsistent coding, staff adoption can take longer, especially in organizations onboarding multiple clinicians or rotating surgical support teams. Lost time may not appear on the purchase order, but it affects efficiency.
Common sourcing mistakes to avoid
- Approving kits without a full component list and replacement list
- Comparing quotations that do not use the same procedural scope
- Ignoring post-sterilization durability and maintenance guidance
- Assuming local distributor stock equals spare-part readiness
- Skipping multi-department review before large-volume rollout
Each of these mistakes can seem small during negotiation, but together they create higher downstream risk. Procurement teams that standardize pre-order review usually experience fewer urgent exceptions, cleaner vendor scorecards, and stronger internal confidence in sourcing decisions.
How strategic buyers reduce risk
Leading procurement teams treat dental implant kits as part of a broader healthcare technology supply strategy. That means validating not only immediate fit, but also continuity, documentation quality, and service responsiveness. In practice, this often includes a dual-source review, annual supplier performance checks, and quarterly spare-parts forecasting.
For organizations operating in cross-border or multi-clinic environments, those disciplines matter even more. Better sourcing outcomes usually come from disciplined technical review, not from faster ordering.
Final Buying Guidance for Procurement Teams
Before ordering dental implant kits, procurement teams should confirm 3 essentials: exact system compatibility, complete documentation, and reliable replacement support. Once those basics are covered, cost comparison becomes more meaningful because buyers are evaluating equivalent clinical and operational value rather than incomplete offers.
For procurement leaders sourcing in healthcare technology markets, the strongest decisions come from balancing technical precision with supply chain discipline. Dental implant kits that appear similar in a catalog can perform very differently once introduced into sterilization, surgery, and replenishment workflows.
TradeNexus Pro helps procurement teams and B2B decision-makers assess high-impact sourcing categories with deeper market clarity, structured evaluation logic, and supplier-facing insight. If you are reviewing dental implant kits for a new tender, private-label project, or multi-site supply program, contact us to discuss your sourcing priorities, request tailored procurement intelligence, or learn more solutions for healthcare technology purchasing.
Get weekly intelligence in your inbox.
No noise. No sponsored content. Pure intelligence.