IV Fluids Equipment Failures That Can Disrupt Ward Workflow
When iv fluids equipment fails, the disruption rarely stays isolated to one pump, one patient, or one room. A stalled infusion, an alarm cascade, or a pressure inconsistency can quickly delay medication delivery, interrupt handoffs, increase manual checks, and slow bed turnover across the ward. In healthcare environments where timing, traceability, and coordination matter every minute, understanding the most common failure points in iv fluids equipment is essential for keeping workflow stable, protecting patient safety, and reducing preventable downtime.
What counts as an IV fluids equipment failure in real ward operations?
In practice, iv fluids equipment failure is broader than a device that stops working completely. It includes any condition that prevents fluid therapy from being delivered accurately, continuously, or safely. That may involve infusion pumps, IV stands, pressure bags, tubing sets, connectors, flow regulators, battery systems, alarm modules, or software-linked monitoring components. Even a small defect in one part of the delivery chain can trigger delays that ripple through nursing rounds, patient transport, and treatment schedules.
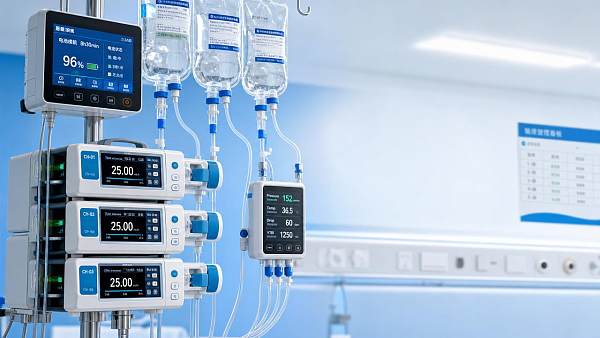
Common examples include unexplained occlusion alarms, inaccurate flow rates, cracked housings, depleted batteries, kinked tubing, sensor failures, loose pole mounts, and calibration drift. In many wards, the operational burden comes not only from the technical fault itself but from the extra verification steps that follow. Staff may need to locate replacement iv fluids equipment, document the event, reassess the patient, restart the infusion, and confirm that no therapy window was missed.
It is also important to distinguish between visible breakdowns and hidden performance failures. A pump that powers on but delivers inconsistent rates can be more disruptive than a unit that fails outright, because the issue may only be discovered after repeated alarm events or delayed therapeutic response. For this reason, ward workflow planning should treat iv fluids equipment reliability as both a maintenance issue and a process control issue.
Which failure points most often disrupt workflow the fastest?
The most disruptive faults are usually the ones that demand immediate staff intervention. Battery failure is a leading example, especially when devices are moved between rooms, imaging areas, or temporary treatment spaces without proper charging cycles. A pump shutting down mid-transfer forces urgent replacement and creates avoidable treatment interruptions.
Another major issue is false or repeated alarm activation. Occlusion alarms caused by tubing misalignment, sensor wear, or incompatible disposables may sound minor, but repeated alarm fatigue drains attention and increases response time. Over the course of a shift, several malfunctioning iv fluids equipment units can absorb a substantial amount of nursing time.
Mechanical wear also plays a direct role. Damaged clamps, unstable poles, poor wheel movement, and connector leakage all affect setup speed and bedside safety. These failures may not require biomedical escalation at first, yet they slow routine tasks and raise the risk of workarounds. When staff begin improvising with tape, repositioned lines, or borrowed accessories, ward consistency suffers.
The fastest workflow disruptions often come from five recurring categories:
- Power and battery instability during patient movement
- Sensor or alarm errors that trigger repeated manual checks
- Tubing and connector issues that stop or slow flow
- Calibration drift affecting infusion accuracy
- Physical wear that makes iv fluids equipment difficult to mount, move, or clean
When these problems occur together in high-occupancy periods, the result is often a chain reaction: delayed infusion start times, slower discharge prep, increased documentation load, and reduced equipment availability for incoming patients.
How can teams tell whether the issue is equipment-related or process-related?
Not every interruption involving iv fluids equipment is caused by the device alone. Some failures are rooted in workflow design, storage conditions, training gaps, or incompatible consumables. A useful first step is to look for patterns. If the same alarm occurs across different operators but only on one model, the cause is likely technical. If the same issue appears across multiple brands during shift change or rapid admissions, process pressure may be the primary factor.
Environmental conditions matter as well. Equipment stored with low batteries, transported without protective handling, or cleaned with unsuitable agents can deteriorate faster. Likewise, inconsistent setup protocols may create avoidable faults that look like hardware defects. For example, poorly routed tubing can cause intermittent occlusion alarms, while rushed priming can produce flow interruptions that mimic pump problems.
A practical way to separate causes is to review incidents using three filters: device condition, consumable compatibility, and workflow timing. This method helps identify whether iv fluids equipment needs servicing, whether accessory selection is creating instability, or whether ward pressure is increasing handling errors.
What should be considered when evaluating or replacing IV fluids equipment?
When recurring faults begin to affect throughput, replacement decisions should focus on operational fit rather than purchase price alone. Reliable iv fluids equipment should support the actual ward environment: frequent movement, repeated cleaning, fast setup, varied acuity, and long operating hours. A unit that performs well in specification sheets but struggles under high-use conditions may become costly through downtime, staff burden, and service interruptions.
Key evaluation criteria include battery endurance, alarm clarity, compatibility with approved disposables, ease of cleaning, serviceability, and preventive maintenance intervals. Digital traceability is increasingly important as well. Equipment that provides usable logs for alarm history, battery status, or infusion events makes troubleshooting faster and supports stronger quality review.
It is equally valuable to assess accessory ecosystems. Some iv fluids equipment platforms depend on proprietary consumables or specialized mounts. While this can improve consistency, it may also create supply constraints if stock planning is weak. In mixed-device environments, standardization often reduces training friction and lowers the risk of setup errors between shifts or departments.
Before replacement or upgrade, review these questions:
- Does the current failure rate justify replacement over repair?
- Are maintenance records showing repeat faults on the same device family?
- Can the new iv fluids equipment integrate smoothly with existing protocols and consumables?
- Will standardization reduce alarm errors, training time, or spare unit demand?
- Are there hidden lifecycle costs in batteries, accessories, or calibration services?
How can wards reduce failure-related downtime before it becomes a patient flow problem?
The most effective response is prevention built into daily operations. Instead of treating iv fluids equipment issues as isolated service calls, wards benefit from readiness routines that catch faults before the next bedside setup. This includes battery checks at shift transitions, visual inspections for tubing path integrity, quick-clean verification, and removal of units with recurring alarm patterns.
Simple governance changes can have a strong effect. A color-coded status process for ready, in-use, under-review, and out-of-service devices helps prevent uncertain units from circulating back into active care areas. Standardized accessory kits also reduce setup variation and lower the chance that incompatible components will be attached to iv fluids equipment in a rush.
Training should be targeted, not generic. If incident records show that most disruptions come from alarm handling, battery management, or tubing placement, then micro-training on those exact issues will outperform broad annual refreshers. Data from service logs, alarm reports, and ward incident notes should feed directly into these updates.
A practical prevention framework often includes:
- Routine pre-use inspection of pumps, poles, clamps, cables, and battery charge
- Scheduled calibration and preventive servicing based on actual utilization
- Standardized consumables matched to approved iv fluids equipment
- Fast quarantine of devices with repeated alarms or intermittent faults
- Feedback loops between ward staff, clinical engineering, and supply planning
What are the most common mistakes when managing IV fluids equipment failures?
One common mistake is normalizing small faults. A loose pole lock, noisy alarm key, or battery with shortened runtime may seem manageable until several units behave the same way under peak demand. Another mistake is focusing only on repair turnaround without examining root causes. If the same iv fluids equipment returns to service repeatedly with the same issue, the real problem may involve cleaning practice, transport damage, or accessory misuse.
A further risk is fragmented ownership. When maintenance, ward operations, and supply teams each hold part of the picture but no shared review process exists, recurring disruption stays invisible for too long. Reliable performance depends on connecting technical data with bedside workflow observations. The goal is not simply to fix broken iv fluids equipment, but to reduce the number of situations where failures interrupt care delivery in the first place.
For organizations seeking stronger visibility into healthcare technology reliability, broader market intelligence can also support better planning. TradeNexus Pro tracks operational and supply-side developments across healthcare technology, helping decision frameworks stay aligned with equipment trends, service expectations, and sourcing risk factors that influence long-term uptime.
How should the next improvement step be prioritized?
Start with the disruptions that consume the most time, not just the failures that look most serious on paper. Review alarm frequency, replacement turnaround, battery-related interruptions, and repeated setup delays by location and shift. Then map which iv fluids equipment issues are technical, which are procedural, and which are tied to consumable or storage inconsistency. That prioritization makes corrective action faster and more defensible.
In most wards, better uptime comes from a combination of three actions: standardizing approved devices and accessories, tightening preventive maintenance around high-use units, and using incident patterns to improve training. When iv fluids equipment is managed as part of total ward workflow rather than as a standalone asset category, the gains extend beyond reliability to smoother treatment continuity, clearer accountability, and more resilient day-to-day operations.
Get weekly intelligence in your inbox.
No noise. No sponsored content. Pure intelligence.