India BIS Updates Medical Supplies Packaging Standard: Biodegradability Mandatory
On May 2, 2026, the Bureau of Indian Standards (BIS) released IS 17872:2026 — a revised standard mandating biodegradability certification for packaging of imported medical supplies, including disposable gloves, dressing kits, and sampling swabs. This development directly affects medical device exporters, packaging suppliers, and logistics stakeholders serving the Indian market — and signals a tightening of environmental compliance requirements at India’s import gate.
Event Overview
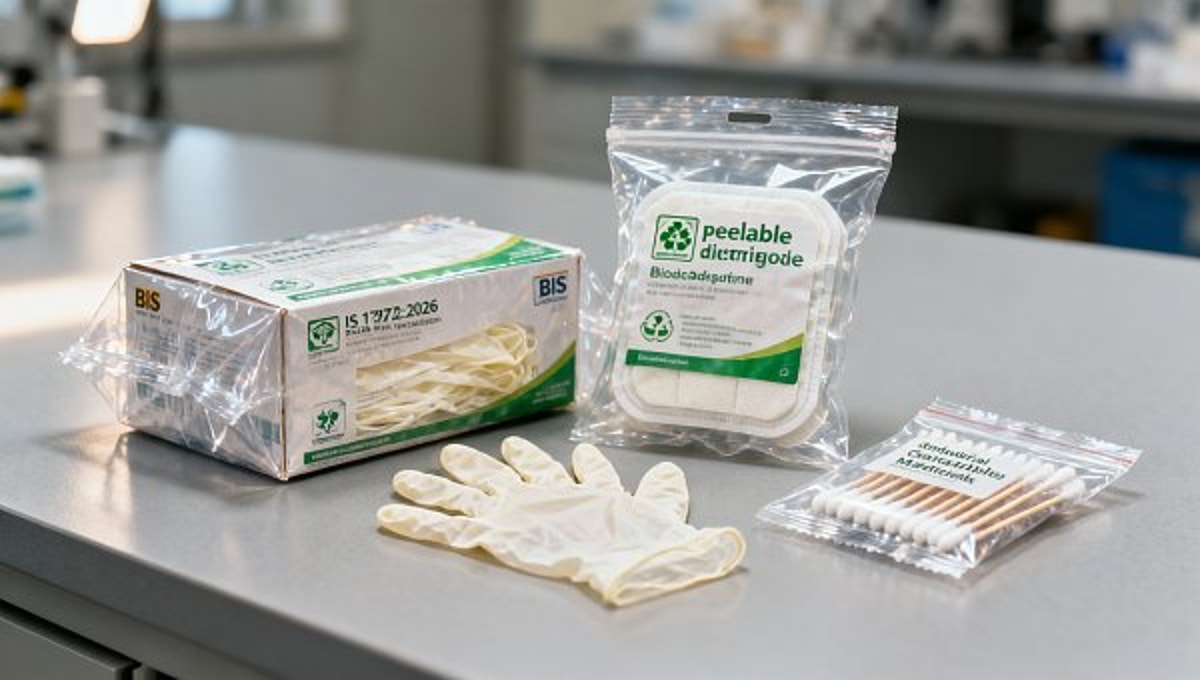
On May 2, 2026, the Bureau of Indian Standards (BIS) published IS 17872:2026, revising the packaging standard for medical supplies. The revision requires that all outer packaging and inner lining materials used for imported medical supplies — specifically disposable medical gloves, sterile dressing kits, and sample collection swabs — must comply with either ASTM D6400 or ISO 17088 for industrial compostability. Certified packaging must bear the official BIS biodegradability mark. Enforcement begins August 1, 2026. During the transition period (May 2 – July 31, 2026), non-certified packaging will incur a 5% environmental surcharge.
Which Subsectors Are Affected
Direct Exporters of Medical Consumables
Exporters shipping disposable gloves, dressing kits, or swabs to India face immediate supply-chain implications: packaging is now a regulated component of product conformity, not just a logistical detail. Non-compliant packaging may result in customs delays, surcharge application, or rejection of consignments post-August 2026.
Packaging Material Suppliers & Converters
Suppliers providing printed films, laminates, pouches, or trays for medical packaging must verify whether their current formulations meet ASTM D6400 or ISO 17088. Certification requires third-party testing and documentation traceable to batch-level production — meaning material-grade consistency and supplier transparency become critical.
Contract Manufacturers & OEMs
OEMs outsourcing packaging assembly — especially those using multi-layer composites or barrier-coated substrates — may need to requalify entire packaging lines. Changes may extend beyond film selection to include ink systems, adhesives, and sterilization compatibility — all of which must be reassessed under the new biodegradability requirement.
Logistics & Customs Intermediaries
Freight forwarders and customs brokers handling medical imports into India must now verify packaging compliance documentation (e.g., test reports, BIS mark usage authorization) prior to clearance. Absence of valid certification may trigger verification hold-ups or surcharge assessments during the transition window.
What Relevant Enterprises or Practitioners Should Focus On Now
Monitor official BIS guidance on mark licensing and certification pathways
While IS 17872:2026 specifies the technical requirement, BIS has not yet published detailed procedures for applying the biodegradability mark — including eligibility criteria for foreign manufacturers, authorized testing labs in India or abroad, or validity duration of test reports. Enterprises should track BIS circulars and updates via the official portal (bis.gov.in).
Identify high-volume SKUs and packaging formats requiring urgent review
Not all packaging components carry equal risk. Priority should be given to SKUs with highest export volume to India — particularly single-use glove boxes, blister-packed swabs, and peelable pouches for dressings — as these are explicitly named in the standard and most likely subject to frontline inspection.
Distinguish between policy signal and operational readiness
The 5% environmental surcharge applies only during the transition period and only to non-certified packaging — not to products themselves. This suggests BIS intends to incentivize preparation rather than penalize immediately. However, enforcement rigor post-August 2026 remains unconfirmed; enterprises should treat certification as mandatory, not optional, from day one of implementation.
Initiate packaging material qualification and supplier alignment now
Certification timelines — including sample submission, lab testing, and report issuance — typically require 6–10 weeks. Given the August 1, 2026 enforcement date, procurement and validation cycles for new packaging must begin no later than early June 2026. Concurrently, suppliers must be engaged to provide updated declarations of conformity and batch-specific test data.
Editorial Perspective / Industry Observation
Observably, this revision reflects India’s broader regulatory shift toward embedding environmental accountability into product-level conformity — moving beyond end-of-life disposal policies to upstream packaging design mandates. Analysis shows that IS 17872:2026 is less an isolated update and more a pilot within BIS’s emerging ‘green conformity’ framework, potentially extendable to other regulated health products. From an industry perspective, it functions primarily as a compliance signal: while enforcement mechanisms remain partially undefined, the inclusion of a financial surcharge and explicit timeline confirms enforceability intent. Continued attention is warranted — especially regarding how BIS interprets ‘inner lining materials’, whether multi-material laminates qualify under current test standards, and whether exemptions apply for packaging used exclusively in hospital-controlled waste streams.
This is not yet a fully operational regime — but it is no longer merely a proposal. Enterprises active in India’s medical imports should treat it as a binding requirement with phased rollout discipline.
Conclusion
IS 17872:2026 marks a formal integration of biodegradability verification into India’s medical import control system. Its significance lies not in novelty — similar packaging sustainability clauses exist in EU MDR Annex I — but in its binding scope, defined timeline, and direct linkage to customs treatment. Currently, it is best understood as a structured transition mandate: one that prioritizes verifiable action over declarative commitment, and rewards proactive alignment over reactive compliance.
Source Disclosure
Primary source: Bureau of Indian Standards (BIS), IS 17872:2026 — “Packaging Requirements for Medical Supplies”, published May 2, 2026. Official text available at bis.gov.in.
Note: Implementation guidelines, mark licensing procedures, and list of accredited testing laboratories remain pending publication and are under ongoing observation.
Get weekly intelligence in your inbox.
No noise. No sponsored content. Pure intelligence.




























