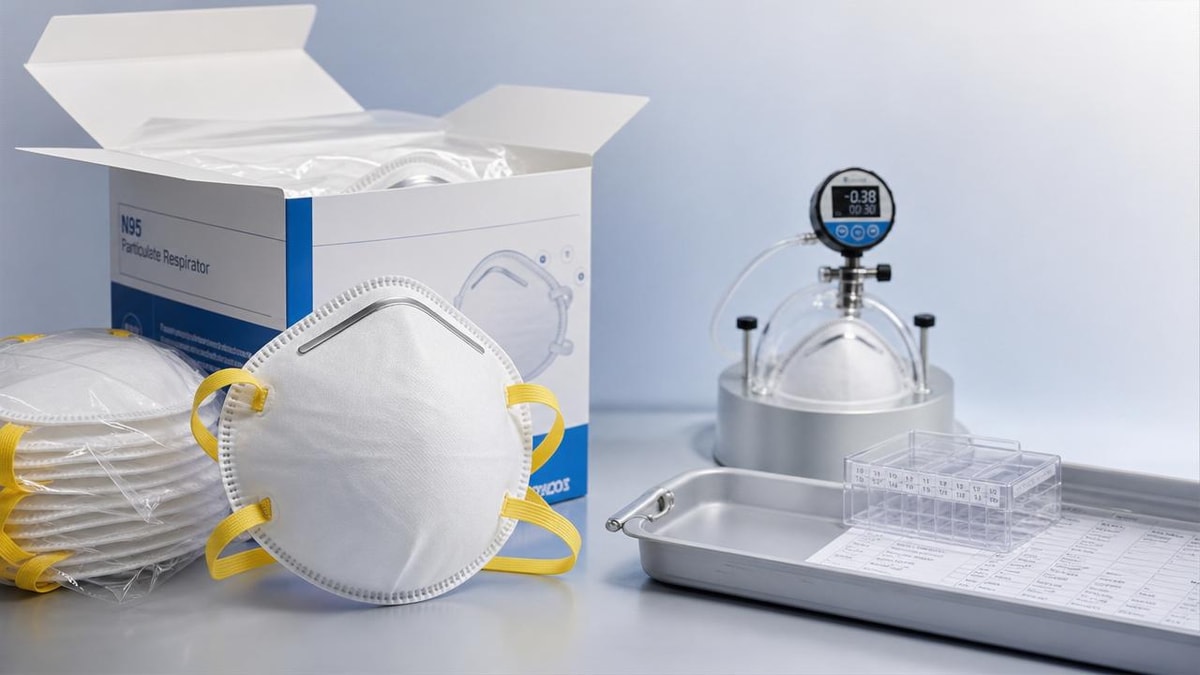
N95 respirators: why fit matters more than many expect
When selecting N95 respirators, filtration efficiency alone is not enough—fit often determines whether real-world protection meets the standard on paper. For quality control and safety managers, understanding seal integrity, user variability, and compliance factors is essential to reducing workplace exposure risks and making better procurement decisions.
A checklist-based approach is the most practical way to evaluate N95 respirators because failure rarely comes from one obvious defect. In real workplaces, protection can be weakened by face shape differences, poor donning habits, incompatible eyewear, facial hair, movement during tasks, storage damage, and incomplete fit testing programs. For quality control teams and safety managers, the goal is not simply to buy compliant products, but to confirm that N95 respirators perform reliably across actual users, actual tasks, and actual operating conditions.
Start here: the first five points to confirm before purchase
Before comparing price, brand reputation, or stated filtration ratings, prioritize the factors that most directly affect protection. This order helps procurement and EHS teams avoid the common mistake of overvaluing laboratory claims while underchecking fit-related performance.
- Confirm the respirator is a legitimate N95 product from a credible manufacturer, with traceable documentation, labeling consistency, and quality records.
- Verify that the design supports a stable facial seal across your workforce, not just a small test group.
- Check whether the model can pass fit testing on the user populations most exposed to airborne hazards.
- Review compatibility with safety glasses, face shields, hearing protection, and task-related movement.
- Assess whether training, storage, replacement intervals, and compliance monitoring are realistic for your site.
This sequence matters because many organizations assume that if a respirator is labeled N95, expected protection is automatic. In practice, an excellent filter with poor face seal performance may deliver less useful protection than a well-fitted model with the same rating. For high-exposure settings, that gap can directly affect incident risk, absenteeism, and regulatory findings.
Core checklist: how to judge whether fit is adequate
When reviewing N95 respirators, use the following fit-focused checklist as a practical screening tool. It helps bridge the gap between product specifications and field use.
- Face seal geometry: Look for a design that conforms well around the nose, cheeks, and chin without obvious pressure gaps. A model that seals on one face shape may fail on another.
- Nose clip performance: The nose area is one of the most common leakage points. Check whether the nosepiece retains shape after repeated adjustments and during long shifts.
- Head strap stability: Straps should maintain tension without slipping, overstretching, or causing users to reposition the respirator frequently.
- Fit during movement: Evaluate sealing while speaking, bending, turning the head, or performing repetitive tasks. Static fit is not enough.
- User comfort: Discomfort often leads to touching, loosening, or incorrect wear. A technically compliant N95 respirator may still underperform if users cannot tolerate it for required durations.
- Fogging indicators: Frequent eyewear fogging can signal leakage around the nose bridge, especially in humid environments.
- Size and model variation: One universal model rarely fits every workforce segment. Multi-model selection usually improves fit test pass rates.
What quality control teams should inspect beyond the datasheet
Datasheets for N95 respirators usually emphasize filtration efficiency, breathing resistance, and certification. These matter, but quality control teams should also inspect build consistency and packaging integrity because fit performance can be affected by manufacturing variation or handling damage.
Physical quality checkpoints
- Uniform weld quality at strap attachment points
- Consistent cup shape or fold structure after unpacking
- No visible deformation from compression during transport
- Stable nose foam or sealing surface materials where applicable
- Clear lot traceability, expiration information, and storage guidance
Even slight deformation can reduce the ability of N95 respirators to seal correctly. If cartons are crushed, humidity is uncontrolled, or storage periods exceed recommendations, fit-related performance may decline before the product ever reaches users.
Documentation checkpoints
Request certification evidence, quality system details, lot control information, and any available fit test support data. For enterprise buyers, supplier transparency is a practical trust signal. It also aligns with stronger sourcing governance, which is increasingly important in global procurement environments such as healthcare technology, advanced manufacturing, and supply chain-led distribution networks.
Fit testing and user variability: the make-or-break factor
The most important reality for safety managers is simple: N95 respirators do not fit all users equally. Face length, nose bridge shape, cheek contour, and chin structure all influence seal quality. This is why formal fit testing is not a paperwork exercise but an operational control.
If your workforce includes mixed demographics, temporary labor, contractors, or multiple shift teams, expect wider fit variation. A single approved model may not be enough. Safety programs that maintain two or three validated N95 respirators often achieve better continuity than those locked into one SKU.
Scenario-based checks for different workplaces
Not every site should judge N95 respirators the same way. The right checklist depends on task profile, contamination risk, and user behavior.
Healthcare and clinical support settings
Prioritize prolonged wear comfort, communication impact, eyewear compatibility, and high-frequency donning procedures. Leakage around the nose bridge and moisture buildup should be monitored closely.
Advanced manufacturing and dusty process areas
Check fit during repetitive head motion, sweating, and coexistence with helmets, goggles, and hearing protection. A respirator that shifts during production tasks creates a hidden exposure pathway.
Warehousing, logistics, and mixed-duty environments
Focus on training simplicity, rapid correct donning, and resistance to handling damage. In these settings, misuse often comes from operational pace rather than technical misunderstanding.
Common blind spots that reduce the value of N95 respirators
Several recurring oversights can make N95 respirators look compliant in procurement records but less effective in the field.
- Assuming certification equals user protection: Certification confirms a standard, not a guaranteed fit on every wearer.
- Ignoring facial hair policies: Even minor facial hair in the sealing area can compromise performance significantly.
- Overlooking training decay: Workers may pass initial instruction but gradually adopt poor donning habits.
- Using only cost as the sourcing filter: Lower-priced options can create hidden costs through lower fit pass rates or more frequent replacement.
- Neglecting inventory rotation: Aged or poorly stored stock may deform and reduce seal reliability.
A practical execution plan for safety managers
If your organization is reviewing or changing N95 respirators, use a staged process rather than a one-step purchase decision.
- Define exposure scenarios and user groups by task, shift, and work area.
- Shortlist qualified N95 respirators from suppliers with strong traceability and documentation.
- Run sample evaluations for construction quality, packaging durability, and comfort.
- Conduct fit testing across representative users, not only supervisors or office-selected volunteers.
- Track fit pass rates, user feedback, and compatibility issues with other PPE.
- Approve more than one model when workforce variability justifies it.
- Train, retrain, and verify user seal check practices on a defined schedule.
This method gives procurement, quality, and safety teams a shared framework. It also supports better vendor conversations because decisions can be tied to measurable field performance instead of marketing claims alone.
FAQ: quick answers buyers and site leaders often need
Are all N95 respirators equally protective if they meet the same standard?
No. N95 respirators may share filtration requirements, but actual protection depends heavily on fit, correct use, and task conditions.
Can user comfort be treated as secondary?
No. Poor comfort often leads to poor compliance, frequent adjustment, and lower real-world protection.
Should one model be deployed sitewide for simplicity?
Only if fit test results support that choice across the workforce. In many operations, multiple approved N95 respirators are the better control strategy.
What to prepare before discussing suppliers or internal approval
To move efficiently from evaluation to implementation, prepare a short decision file covering exposure type, user count, fit test expectations, PPE compatibility needs, wear duration, replacement assumptions, storage conditions, and acceptable documentation standards. For supplier discussions, ask for evidence on lot consistency, lead times, packaging robustness, and support for fit validation programs.
For organizations that rely on strategic sourcing intelligence, a disciplined review of N95 respirators should connect product compliance with operational usability. That is where quality control and safety management create the most value: translating standards into dependable workplace protection.
If you need to confirm model suitability, fit-related risk points, sourcing stability, implementation timelines, or budget trade-offs, the best next step is to align procurement, EHS, and frontline supervisors around the same checklist before final selection. With N95 respirators, the smartest buying decision is usually the one that proves fit in practice, not just performance on paper.
Get weekly intelligence in your inbox.
No noise. No sponsored content. Pure intelligence.