How Accurate Are Portable Ultrasound Scanners?
Portable ultrasound scanners have advanced rapidly, but how accurate are they compared with full-size systems? For buyers, clinicians, and healthcare technology evaluators exploring remote patient monitoring, telemedicine carts, smart glucometers, digital blood pressure monitors, and wearable ecg monitors, accuracy is critical. This article examines real-world performance, diagnostic limits, and procurement factors to help decision-makers assess whether portable ultrasound scanners are fit for clinical, operational, and commercial use.
In B2B healthcare technology purchasing, accuracy is rarely a single-number question. It depends on probe design, imaging mode, operator skill, clinical use case, software processing, network integration, and the tolerance for false negatives or missed findings in a given workflow. A handheld scanner used for point-of-care triage has a very different performance target than a cart-based system used for detailed echocardiography or fetal anomaly screening.
For procurement teams, distributors, project managers, and enterprise decision-makers, the right question is not whether portable ultrasound scanners are “good” or “bad.” The practical question is where they are sufficiently accurate, where they are limited, and what technical and commercial safeguards should be built into evaluation and deployment.
What Accuracy Means in Portable Ultrasound Scanners
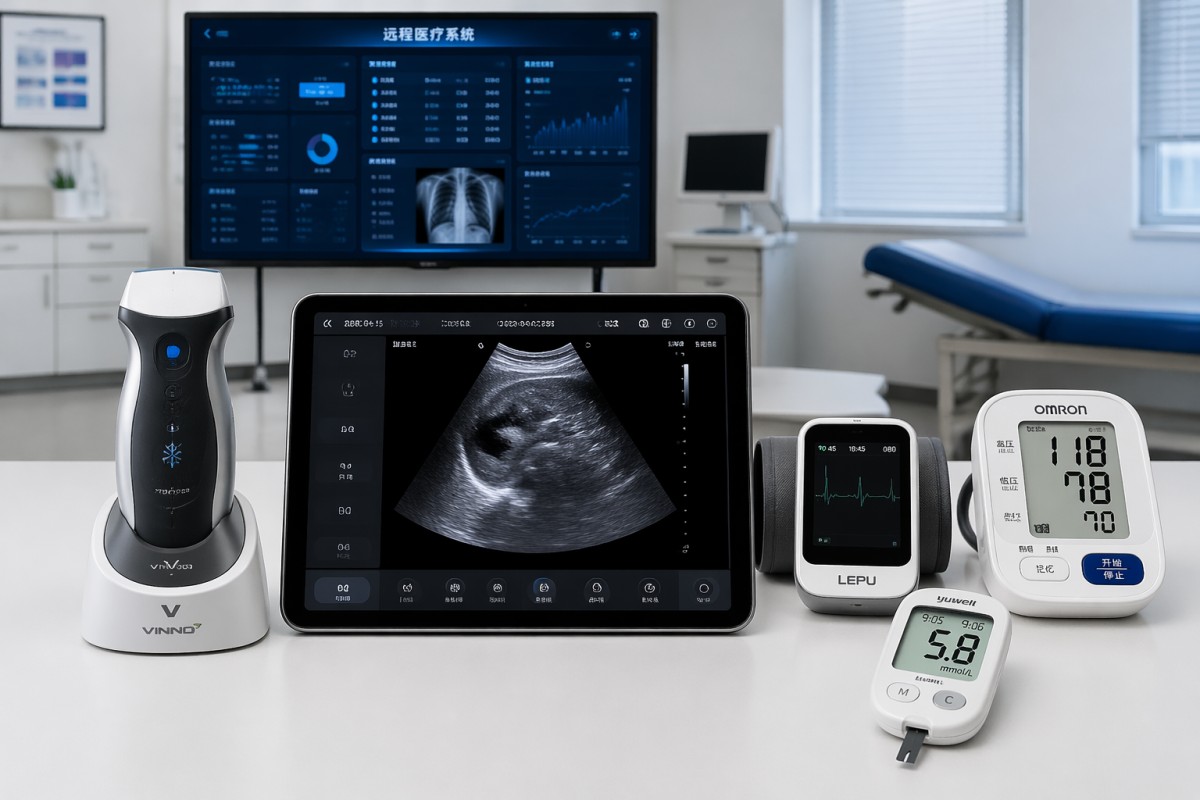
Portable ultrasound scanners can deliver clinically useful imaging in many frontline settings, especially for focused assessments performed at the bedside, in ambulances, rural clinics, outpatient facilities, and telemedicine-supported programs. In many routine point-of-care applications, modern devices provide image quality that is strong enough for rapid decision support within 1–5 minutes of scanning.
However, “accuracy” includes several layers. One is image resolution: how clearly structures can be visualized. Another is diagnostic agreement: whether findings match those from full-size systems or follow-up imaging. A third is reproducibility: whether two trained operators reach similar conclusions using the same portable scanner under similar conditions.
In practice, portable ultrasound scanners are often most accurate when used for binary or focused questions. Examples include detecting free fluid, confirming bladder volume, checking basic cardiac activity, identifying pleural effusion, guiding vascular access, or assessing gross abdominal findings. They are less reliable for complex, highly detailed studies that require advanced Doppler analysis, deep penetration, or prolonged measurements across multiple anatomical planes.
Key variables that shape diagnostic performance
Procurement teams should assess at least 6 variables before making claims about accuracy: transducer frequency range, imaging presets, grayscale processing, Doppler capability, battery stability during long shifts, and interoperability with PACS or cloud review systems. A scanner with a 2–5 MHz phased array probe serves different needs than a 7–15 MHz linear probe, and accuracy must be judged accordingly.
- Focused emergency assessment often values speed and acceptable sensitivity over full-spectrum detail.
- Musculoskeletal and vascular imaging usually require higher-frequency probes and strong near-field resolution.
- Obstetric, cardiac, and complex abdominal imaging may demand broader software tools and more advanced measurement packages.
- Tele-ultrasound workflows need stable data transfer, standardized acquisition protocols, and secure archiving.
This means there is no universal performance score. A portable device may achieve high clinical utility in 3 departments and still be unsuitable for 2 other departments that require more advanced quantification. That distinction matters for both capital approval and risk management.
The table below shows how accuracy expectations typically vary by application rather than by device category alone.
The main takeaway is straightforward: portable ultrasound scanners are often accurate enough for focused, high-speed decision support, but not every model is built for high-complexity diagnostics. Buyers should define the exact diagnostic question set before evaluating performance claims.
How Portable Systems Compare with Full-Size Ultrasound Platforms
Full-size ultrasound systems still lead in image depth, advanced Doppler, thermal management for extended sessions, screen size, workflow customization, and support for longer examinations. In departments that require multi-parameter measurements over 20–45 minutes, fixed or cart-based units retain a clear operational advantage.
That said, the gap has narrowed significantly over the last 5–8 years. Many portable ultrasound scanners now offer high-resolution B-mode imaging, color Doppler, preset-driven workflows, wireless connectivity, and AI-assisted labeling. For frontline screening and procedural support, the difference between portable and full-size units may be less about raw imaging ability and more about consistency under demanding conditions.
A practical evaluation should compare use case by use case. A handheld unit can outperform a full-size machine that is physically unavailable at the point of care. In mobile medicine, home care, emergency transport, and decentralized networks, faster access can improve decision timing even when image sophistication is somewhat lower.
Where portable scanners perform well
Portable systems are commonly effective in 4 high-value conditions: bedside triage, procedural guidance, rural or community outreach, and connected telemedicine workflows. In these scenarios, the operational benefit of immediate imaging may offset moderate limits in advanced quantification.
Where full-size systems remain stronger
Complex cardiac analysis, detailed obstetric examinations, deep abdominal imaging in high-BMI patients, and specialty radiology studies still benefit from larger systems. These tasks may require wider dynamic range, better probe selection, higher frame stability, and stronger measurement packages than entry-level portable devices can deliver.
The comparison table below is useful for sourcing teams balancing capital cost, mobility, and expected clinical output.
For commercial buyers, the implication is clear: portable ultrasound scanners should not be benchmarked only against the technical ceiling of premium consoles. They should be benchmarked against the actual care pathway, user profile, and response-time requirement of the intended service model.
The Main Factors That Influence Accuracy in Real-World Use
Even a well-designed device can underperform if workflow controls are weak. Real-world accuracy depends on more than the scanner itself. Operator training, standard scanning protocols, maintenance, cleaning routines, software updates, and image review governance all affect outcomes. In many deployments, user variation accounts for a large share of inconsistency during the first 30–90 days.
Probe selection is one of the most overlooked variables. A linear probe in the 7–15 MHz range is better suited for superficial vessels, soft tissue, and line placement. A phased array around 2–5 MHz is more practical for cardiac windows and deeper structures. Using the wrong probe can reduce diagnostic confidence even if the hardware itself is high quality.
Environment also matters. Mobile care sites, home visits, emergency vehicles, and pop-up clinics often involve lighting variation, unstable patient positioning, limited privacy, and inconsistent power or charging access. A scanner that tests well in a demo room may behave differently during a 12-hour field shift with repeated cleaning cycles and intermittent connectivity.
Operational controls that improve performance
- Define 5–10 approved use cases rather than allowing unrestricted use across every department.
- Use standardized protocols for image acquisition, labeling, and escalation to full-size imaging.
- Set a training threshold, such as supervised scanning for the first 20–50 examinations per user.
- Review image quality monthly during the first quarter after deployment.
- Track downtime, battery aging, software update frequency, and probe failure trends.
These controls are especially important for enterprise networks introducing portable ultrasound scanners alongside digital blood pressure monitors, smart glucometers, and wearable ECG monitors. Once imaging becomes part of a connected care model, consistency and data governance matter as much as hardware specifications.
Common sources of overestimated accuracy
Overestimation often happens when buyers rely only on vendor demos, compare ideal images rather than average images, or ignore operator variability. Another common issue is testing on low-complexity cases only. A more reliable assessment includes at least 3 user levels, 2 care settings, and a sample of normal and challenging patients.
For quality and safety managers, escalation criteria should be documented in advance. If image clarity is inadequate, if measurements fall outside expected range, or if a high-risk finding is suspected, users should move to confirmatory imaging rather than extending bedside scanning beyond its intended scope.
How Buyers Should Evaluate Portable Ultrasound Scanners Before Procurement
A solid procurement process should combine clinical validation with commercial review. Cost alone is a weak selection criterion because a lower-priced scanner may generate higher downstream expense through repeat imaging, workflow delays, or insufficient interoperability. Buyers should evaluate total deployment value over 3–5 years, not just initial unit price.
At minimum, sourcing teams should review image quality, supported probes, software features, battery runtime, charging cycle practicality, disinfection compatibility, data export options, cybersecurity posture, user licensing, warranty structure, and service response time. For cross-border B2B transactions, spare parts lead times and local training availability are also important.
Pilot testing is strongly recommended. A 2–6 week evaluation period often gives a clearer picture than a one-day demonstration. During that pilot, organizations should compare scan completion rates, number of escalations to conventional imaging, clinician satisfaction, and usable image percentage across shifts and user types.
A practical procurement checklist
The table below can help procurement directors, financial approvers, and project leads evaluate whether a portable ultrasound scanner is suitable for clinical and commercial rollout.
A scanner that scores well across all 4 areas is usually a better strategic investment than a device selected on headline specifications alone. For distributors and channel partners, these same criteria also improve post-sale customer retention and reduce support disputes.
Questions financial approvers should ask
- Will the scanner reduce referral delay, transport time, or duplicated imaging in measurable workflows?
- How many users need onboarding, and is training included in the purchase scope?
- What is the expected refresh cycle: 3 years, 5 years, or longer?
- How quickly can service support or replacement units be delivered in the target region?
These questions link diagnostic accuracy to operational ROI, which is essential in enterprise healthcare technology decisions.
Best-Fit Use Cases, Limits, and Frequently Asked Questions
Portable ultrasound scanners are best viewed as workflow enablers with defined clinical boundaries. They can be highly valuable in urgent care, home health, critical access facilities, mobile screening programs, and decentralized outpatient networks. In these settings, access to imaging within minutes can improve triage, procedural confidence, and care coordination.
Their limits are equally important. If a service line requires highly reproducible measurements, advanced spectral Doppler, or deep-tissue detail across complex anatomy, buyers should plan for a hybrid model. That may include handheld scanners for first-line screening and full-size systems for follow-up or specialist review.
For healthcare technology portfolios that already include telemedicine carts, connected monitoring devices, and cloud-based care platforms, portable ultrasound scanners can extend decision support at the edge. The key is disciplined scope definition, training, and escalation.
FAQ: Are portable ultrasound scanners accurate enough for cardiac exams?
They can be accurate enough for focused cardiac questions such as gross ventricular activity, pericardial effusion screening, or basic hemodynamic impression. They are less suitable for comprehensive echocardiography requiring advanced measurements, detailed valve analysis, or specialist-grade repeatability.
FAQ: How long does implementation usually take?
A typical enterprise rollout may take 2–8 weeks depending on device quantity, user count, integration needs, and training depth. A small pilot for 1 department can move faster, while a multi-site deployment with cloud workflow and governance approvals often takes longer.
FAQ: What are the biggest mistakes buyers make?
Three common mistakes are buying for too many use cases at once, ignoring service and integration requirements, and assuming demo-room image quality will match field conditions. Another risk is failing to define who can scan, when to escalate, and how image quality will be reviewed over time.
FAQ: Which teams benefit most from portable systems?
Emergency care teams, critical care units, mobile clinicians, vascular access specialists, community health programs, and telemedicine-enabled networks often see the fastest operational value. Procurement, quality, and finance teams also benefit when the deployment reduces avoidable transfers, repeat scans, or workflow bottlenecks.
Portable ultrasound scanners are no longer niche devices. When matched to the right clinical task, they can provide dependable accuracy, faster imaging access, and stronger mobility than traditional systems. When misapplied, they can create false confidence, repeated work, and avoidable risk. The difference lies in disciplined selection and implementation.
For organizations evaluating healthcare technology portfolios across remote monitoring, telemedicine infrastructure, and connected diagnostics, portable ultrasound scanners can be a strong addition when accuracy is judged by real-world use case rather than marketing claims. TradeNexus Pro supports B2B buyers, channel partners, and decision-makers with practical insight into selection criteria, deployment readiness, and strategic sourcing. To explore tailored solutions, compare sourcing options, or discuss commercial fit, contact us to get a customized plan and learn more about the right portable imaging pathway for your business.
Get weekly intelligence in your inbox.
No noise. No sponsored content. Pure intelligence.



























