How to Choose MRI Machine Components in 2026
Choosing the right MRI machine components in 2026 means balancing imaging performance, compliance, lifecycle cost, and supply stability. For buyers and technical teams comparing healthcare technology options alongside portable ultrasound scanners, smart glucometers, digital blood pressure monitors, wearable ECG monitors, remote patient monitoring systems, and telemedicine carts, this guide outlines the critical factors, sourcing priorities, and evaluation criteria needed to make smarter procurement and deployment decisions.
What MRI machine components matter most in 2026 procurement?
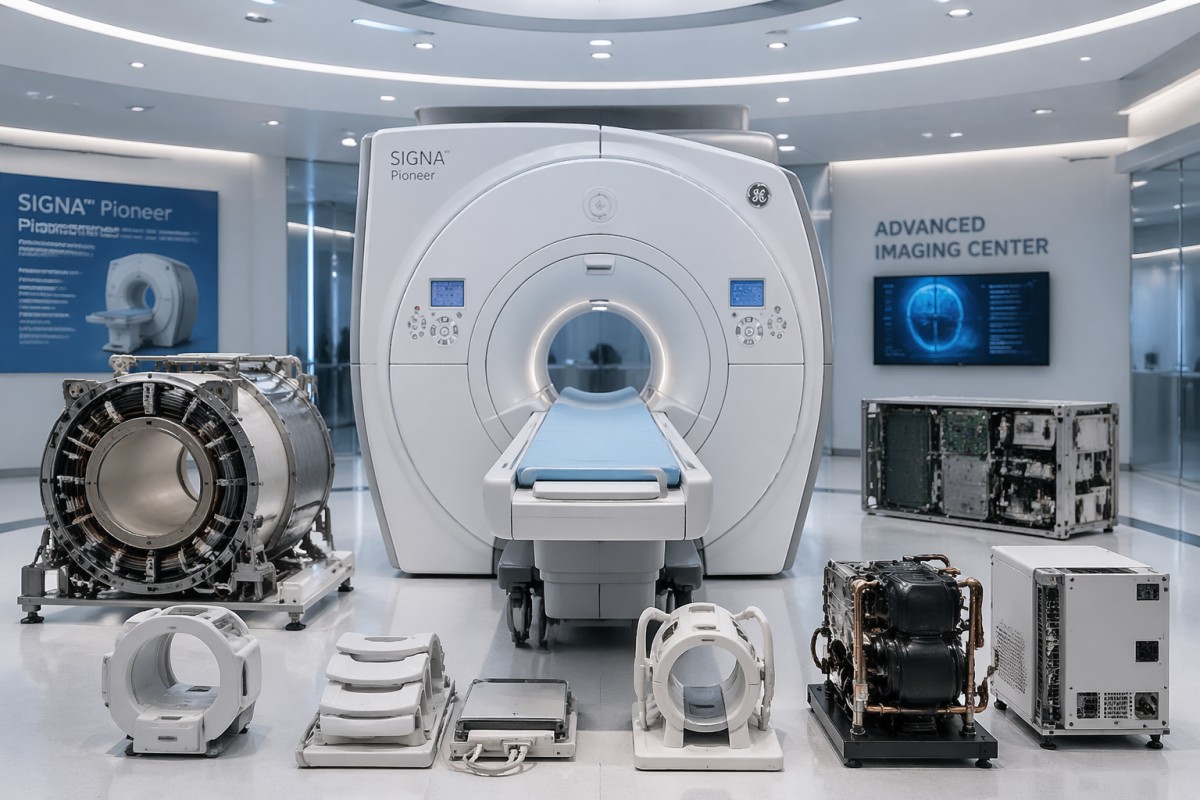
MRI machine components are not purchased as isolated parts. In practice, procurement teams evaluate a connected system that includes the magnet assembly, gradient coils, RF coils, patient table, cooling subsystem, power electronics, shielding interfaces, software controls, and service support. In 2026, the pressure is higher because hospitals and imaging networks want stable uptime, lower total ownership cost, and clearer compatibility planning over a 5–10 year lifecycle.
For operators and project managers, component choice directly affects image consistency, scan throughput, maintenance intervals, and workflow reliability. For finance and business reviewers, the question is broader: does the component strategy reduce unplanned replacement risk, shorten service lead time from 2–8 weeks to a manageable window, and protect budget approval from hidden aftermarket costs? These are now core selection issues, not secondary technical details.
Procurement decisions also sit inside a larger healthcare technology stack. A facility that is investing in MRI upgrades may also be standardizing remote monitoring, connected diagnostics, and digital care infrastructure. That makes interoperability, supplier responsiveness, and documentation quality more important than a narrow parts price comparison.
Core component groups buyers should map first
Before comparing quotations, create a component map with 4 layers: field generation, signal acquisition, patient handling, and system support. This simple structure helps technical and commercial teams speak the same language during vendor evaluation and prevents critical omissions in tenders.
- Field generation: magnet, cryogenic support where applicable, gradient subsystem, and associated power modules.
- Signal acquisition: RF transmit and receive coils, preamplifiers, connectors, and shielding-related interfaces.
- Patient handling: table, positioning hardware, motion-related accessories, and safety interlocks.
- System support: software control boards, cooling units, cabling, spare kits, documentation, and field service coverage.
This structure is especially useful for distributors, agents, and enterprise sourcing teams that manage multiple facilities. It supports cleaner RFQ documents, more accurate replacement planning, and a faster technical clarification cycle, often reducing internal review rounds from 3–4 meetings to 1–2 focused approval sessions.
How should buyers compare MRI machine components by performance and replacement risk?
A good comparison framework should separate mission-critical performance from service-critical risk. Not every component has the same impact on image quality or downtime. A gradient amplifier issue can affect scan performance immediately, while a worn patient table assembly may create workflow and safety concerns before image degradation appears. Buyers should rank components by clinical impact, failure consequence, and sourcing complexity.
In 2026, many organizations are moving away from price-only replacement sourcing. They are instead comparing component options against 5 core dimensions: compatibility, expected service life, maintenance burden, lead time, and documentation completeness. This is particularly important when evaluating OEM-aligned parts against third-party or refurbished alternatives.
The table below gives a practical comparison view for MRI machine components often discussed during upgrade, repair, or spare-part planning. It is designed for procurement teams, quality managers, and engineering leads who need a structured review tool.
This comparison shows why MRI machine components should be evaluated with different weightings. A lower-cost RF coil may look attractive, but if it increases retakes or fails cleaning and handling expectations, its real cost rises quickly. Likewise, a power or cooling component with weak documentation can create weeks of avoidable service delay.
A practical ranking method for mixed stakeholder teams
Use a 100-point weighted scorecard split across technical fit, compliance evidence, delivery reliability, and lifecycle support. Many organizations use a range such as 35 points for technical fit, 25 for compliance and traceability, 20 for service and spares, and 20 for commercial terms. The exact weighting can change, but the method creates consistency across bids.
Questions worth asking before approval
- Does the supplier provide serial-level or model-level compatibility evidence rather than generic claims?
- Is the expected lead time stable at 7–21 days, or does it fluctuate to 6–8 weeks during peak demand?
- Are installation instructions, maintenance records, and replacement conditions clearly documented?
- Can the component be supported with spare access and technical response over the next 3–5 years?
Platforms like TradeNexus Pro help procurement leaders move beyond fragmented vendor claims by organizing supply-side intelligence, technical context, and market signals into a more decision-ready view. That matters when buyers need not only a part, but a dependable sourcing pathway.
Which technical specifications and compliance checks should not be skipped?
The most common sourcing mistake is treating MRI machine components as simple interchangeable hardware. In reality, even small differences in connector type, thermal capacity, software compatibility, or shielding requirements can affect installation success. Technical teams should document baseline specifications before vendor outreach, especially for replacement programs spread across multiple sites.
Compliance review is equally important. Buyers should verify applicable documentation linked to medical device use, electrical safety, electromagnetic considerations, and quality-system traceability where relevant. The exact paperwork depends on the component and market, but the principle is consistent: procurement should confirm what is required for import, installation, hospital acceptance, and service recordkeeping before a purchase order is released.
The next table summarizes common checkpoints used by healthcare technology procurement teams when screening MRI machine components. It is especially useful for quality control managers and safety officers who need a repeatable validation process.
A structured compliance check like this is not bureaucratic overhead. It reduces the chance of project delays, disputed acceptance, and emergency re-sourcing. For cross-border trade, even 1 missing document set can push delivery and commissioning back by 1–3 weeks.
Technical parameters that usually deserve deeper review
Not every project needs the same level of specification depth. However, three areas repeatedly influence sourcing outcomes: electrical and thermal performance, mechanical fit and load tolerance, and interface or software compatibility. Teams should capture these early, not after quotation comparison.
Minimum technical review checklist
- Confirm component generation, interface version, and system-side dependencies before requesting price.
- Review environmental and handling conditions, including storage and transit expectations where sensitive parts are involved.
- Validate installation requirements, from calibration support to site readiness and acceptance testing steps.
- Agree on post-delivery response terms, including technical support windows and replacement handling if mismatch occurs.
For many procurement teams, this checklist shortens technical clarification and helps finance reviewers understand why one quotation carries less operational risk than another, even when the list price is higher.
How do cost, service, and alternatives affect the final buying decision?
MRI machine component cost should be evaluated across the full ownership window, not just at invoice level. A lower purchase price may still be the more expensive decision if installation support is weak, returns are difficult, or delivery uncertainty causes scanner downtime. For imaging centers with dense scheduling, even 2–3 lost operating days can outweigh a visible upfront saving.
Buyers generally compare three routes: OEM-linked replacement, verified third-party supply, and refurbished or remanufactured options. Each route has a place, but the right choice depends on component criticality, site risk tolerance, and available engineering support. Clinical-use intensity also matters. A high-throughput urban imaging center may prioritize service continuity over initial discount, while a lower-volume site may accept a broader alternative range.
A useful procurement approach is to split components into Tier 1, Tier 2, and Tier 3 based on operational consequence. Tier 1 parts affect image quality or core uptime and often justify stricter sourcing. Tier 2 parts support workflow and safety. Tier 3 items may allow more flexibility on aftermarket sourcing if documentation and traceability are still acceptable.
Where alternative sourcing can work, and where it often fails
Alternative sourcing can be effective when the component has stable interface requirements, clear service history, and lower integration complexity. It becomes risky when compatibility depends on firmware, calibration behavior, high thermal load, or site-specific tuning. In those cases, the cheapest option often creates the highest total project burden.
- Better candidates for broader sourcing review: selected accessories, some table-related parts, standardized cables, and non-core support assemblies with clear specifications.
- Higher-risk candidates needing stricter review: gradient-related assemblies, sensitive RF components, control electronics, and cooling modules with tight integration requirements.
- Commercial checkpoint: compare not only unit price, but warranty terms, replacement window, logistics responsibility, and technical response time within 24–72 hours.
TradeNexus Pro is particularly valuable here because purchasing teams can monitor supplier positioning, sector-specific trends, and cross-category technology signals in one place. That wider view helps enterprises avoid buying decisions that solve a short-term repair need but create a longer-term supply bottleneck.
Implementation timeline buyers should expect
For planned component replacement, many projects follow a 4-step path: requirement capture, technical validation, commercial negotiation, and delivery plus acceptance. If data and documents are complete, the internal cycle may take 1–3 weeks. Cross-border orders or multi-site approvals can extend this to 4–8 weeks, especially when installation coordination is required.
Project managers should build time for three common friction points: model confirmation, logistics packaging review, and acceptance documentation. These sound minor, but they often decide whether a component arrives ready for deployment or sits idle waiting for clarification.
What mistakes do buyers make when choosing MRI machine components?
One common mistake is assuming all MRI machine components with similar descriptions are functionally equivalent. In reality, naming conventions can be broad while interface and performance details remain highly specific. Buyers who approve orders from generic descriptions alone often face rework, delayed installation, or incomplete accessory sets.
Another mistake is separating procurement from operator feedback. Technologists and service engineers often identify practical problems earlier than purchasing teams do: cable routing issues, handling difficulty, cleaning limitations, or setup inefficiencies that do not appear on a quotation sheet. Their input can prevent poor choices with very little added review time.
A third issue is underestimating documentation quality. When replacement components are sourced quickly, teams may defer service records, compatibility notes, or transit handling checks. This can create acceptance disputes later, especially in organizations with strict quality or safety management procedures.
FAQ for procurement, engineering, and distribution teams
How should we choose between OEM, third-party, and refurbished MRI machine components?
Start with clinical risk and downtime tolerance. For core components affecting image quality or scanner availability, stricter sourcing is usually justified. For lower-risk assemblies, verified third-party or refurbished routes may be acceptable if compatibility, traceability, and support terms are clear. Use a side-by-side review of 4 dimensions: technical fit, service availability, lead time, and commercial exposure.
What lead time is typical for MRI machine components?
It varies by component and region. Readily available accessories or common support parts may move in 7–15 days, while specialized assemblies can require 3–8 weeks. Cross-border shipments, export checks, or multi-party technical validation can extend schedules further. Buyers should always request an estimated dispatch range plus packaging and acceptance details.
What should quality and safety teams verify before approval?
They should confirm compatibility evidence, traceability, handling conditions, labeling, service or inspection records where relevant, and installation or acceptance requirements. If the component is safety-sensitive or operationally critical, teams should also review return handling and post-installation support responsibilities before final sign-off.
Can MRI sourcing be evaluated alongside other healthcare technology categories?
Yes, and that is increasingly common. Enterprise buyers often assess MRI machine components within a wider portfolio that includes ultrasound, connected monitoring, digital diagnostics, and telemedicine equipment. This allows better supplier consolidation, more coherent service planning, and clearer capital allocation across 12–24 month procurement cycles.
Why work with TradeNexus Pro when evaluating MRI machine components?
For serious B2B buyers, the challenge is rarely just finding a component name. The real challenge is filtering technical claims, identifying credible supply options, understanding lifecycle implications, and matching sourcing decisions to business priorities. TradeNexus Pro supports that process with sector-focused market intelligence, procurement-oriented analysis, and structured visibility across healthcare technology and adjacent strategic industries.
This matters for procurement directors, supply chain managers, engineering project leaders, and distributors who need more than fragmented listings. They need context: which component categories are becoming harder to source, which evaluation criteria deserve stricter scrutiny, and how technology decisions connect to larger capital planning and service resilience goals over the next 3–5 years.
If you are comparing MRI machine components in 2026, TradeNexus Pro can help you structure the decision before budget is committed. That includes parameter confirmation, sourcing route assessment, supplier comparison logic, documentation priorities, lead-time planning, and multi-category procurement alignment.
What you can discuss with our team
- Component selection criteria for RF coils, support assemblies, cooling modules, and replacement planning by site type.
- Quotation comparison frameworks covering compatibility, lifecycle cost, service conditions, and delivery risk.
- Procurement timelines, documentation checkpoints, and cross-border supply considerations for hospital or distributor projects.
- Broader healthcare technology sourcing strategy across imaging, diagnostics, remote monitoring, and telemedicine infrastructure.
If your team is preparing a shortlist, validating specifications, checking certification expectations, requesting sample support where applicable, or aligning internal approval for a formal quote, connect with TradeNexus Pro. A more structured evaluation process reduces avoidable risk and helps turn component sourcing into a stronger procurement decision.
Get weekly intelligence in your inbox.
No noise. No sponsored content. Pure intelligence.



























