Are Smart Glucometers Worth the Extra Cost?
As connected care expands, many buyers are asking whether smart glucometers truly justify their higher price compared with standard devices. For healthcare teams, distributors, and procurement leaders evaluating smart glucometers alongside digital blood pressure monitors, wearable ecg monitors, and broader remote patient monitoring tools, the real question is not just cost—but data accuracy, workflow efficiency, patient adherence, and long-term value.
In B2B healthcare technology purchasing, a glucometer is no longer just a single-use device. It can be a data endpoint, a workflow tool, and a component in a wider remote patient monitoring strategy. That shift changes how hospitals, clinics, distributors, and enterprise buyers should evaluate price. A device that costs 20% to 60% more upfront may still lower total operating cost if it reduces manual logging, improves patient follow-up, or supports multi-device integration.
For procurement teams and business evaluators, the answer depends on use case. A low-cost standard meter may be sufficient for high-volume spot checks in basic care settings. A smart glucometer becomes more compelling when organizations need app connectivity, trend visualization, cloud transfer, caregiver alerts, or easier data sharing across 30-day, 60-day, or 90-day monitoring periods. The right choice is less about headline price and more about measurable use value.
Why Smart Glucometers Are Gaining Attention in Healthcare Procurement
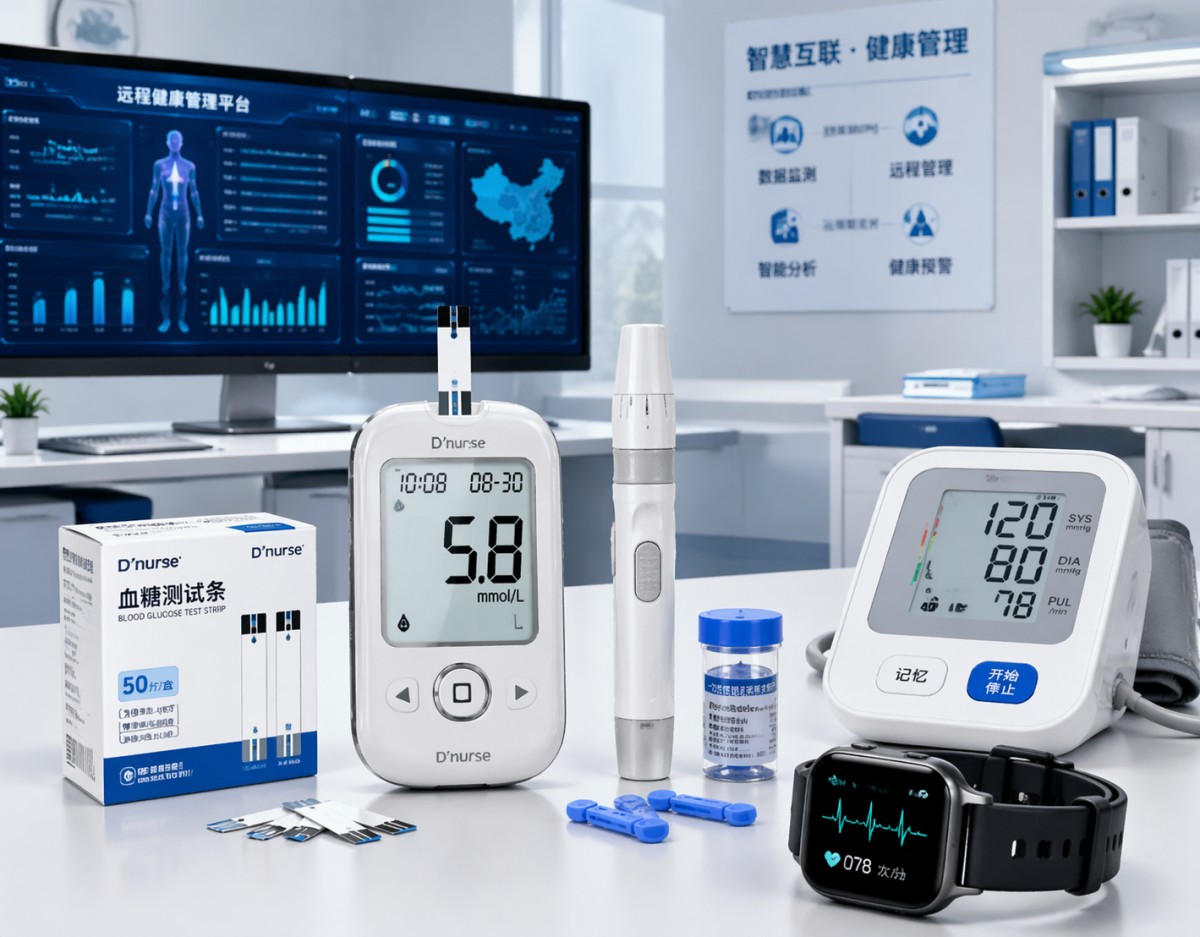
Smart glucometers combine blood glucose testing with digital functions such as Bluetooth sync, mobile app pairing, cloud reporting, timestamped logs, and sometimes integration with broader chronic care platforms. In many healthcare environments, these features address one of the most persistent issues in diabetes management: missing, incomplete, or delayed glucose records. For patients managing type 1, type 2, gestational diabetes, or post-discharge recovery, data continuity matters as much as the individual reading.
From a B2B perspective, demand is rising because glucometers are increasingly evaluated as part of a connected device portfolio. Procurement teams often compare them with digital blood pressure monitors, pulse oximeters, and wearable ECG monitors when building home-care kits, chronic disease programs, or employer health packages. In these scenarios, a smart glucometer can reduce manual handling steps from 4 or 5 down to 2 or 3, especially when readings move automatically into a patient dashboard.
Another reason for market interest is patient adherence. Standard devices depend heavily on users writing down readings or verbally reporting them during a follow-up visit. Smart models may improve adherence by using reminders, trend notifications, and simple visual dashboards. Even when the meter itself is only slightly more advanced, these surrounding digital functions can influence whether care teams receive complete records over a 7-day or 30-day period.
Distributors and enterprise decision-makers should also note that digital capability supports product differentiation. In a crowded device market, the ability to bundle a smart glucometer with software access, patient education, and remote monitoring workflows can create stronger recurring value than selling a standalone commodity device. That is particularly relevant for channels serving private clinics, telehealth providers, and regional healthcare networks.
Core value drivers beyond the device price
- Automatic data capture can reduce transcription errors and save 1 to 3 minutes per patient interaction.
- Digital logs support better trend review over 14-day, 30-day, and 90-day treatment cycles.
- Connected platforms can help care teams prioritize out-of-range readings faster.
- Shared dashboards may improve coordination between patients, caregivers, nurses, and physicians.
The table below shows how standard and smart glucometers differ across practical procurement criteria rather than marketing claims alone.
The key takeaway is simple: smart glucometers are not automatically better for every buyer, but they become economically attractive when data movement, adherence, and remote visibility have operational value. In basic high-volume testing, standard meters remain relevant. In connected care programs, the value equation shifts quickly.
When the Extra Cost Makes Financial and Operational Sense
The most common procurement mistake is comparing device price without calculating workflow cost. In many healthcare settings, labor time, missed follow-up, and incomplete documentation create hidden expense. If a nurse, coordinator, or care manager spends even 2 minutes per patient per week reconciling glucose readings manually, that cost compounds across 100, 500, or 1,000 monitored patients. Over a 12-month program, software-enabled automation may offset a significant portion of the higher hardware price.
Smart glucometers make the most sense in four scenarios: remote patient monitoring, chronic disease programs, discharge-to-home transitions, and multi-site care networks. In each case, timely data movement is more important than the test strip result alone. A meter that uploads readings within seconds can support faster interventions than one that relies on handwritten logs reviewed every 2 to 4 weeks.
Financial approvers should also consider utilization risk. A premium device loses value if patients do not pair it, use the app, or understand alerts. That means the return on investment depends on onboarding quality, interface simplicity, and support materials. A slightly less advanced smart glucometer with easier setup may outperform a feature-rich model that creates friction during the first 7 days of use.
For distributors and channel partners, the extra cost can also be justified by stronger account retention. Buyers are often more willing to renew or expand contracts when the device supports reporting, clinician review, or platform integration. The margin opportunity may come not only from the meter but also from accessories, service support, training, or recurring software-linked programs.
Use cases where smart devices usually outperform basic models
- Telehealth programs that need weekly or daily data visibility.
- Home-care providers managing patients across 2 or more locations.
- Employer or insurer wellness programs tracking adherence over 30 to 90 days.
- Post-discharge monitoring where early intervention can reduce readmission risk.
Cost should be measured in three layers
- Device acquisition cost, including meter, lancets, strips, and accessories.
- Operational cost, including setup, training, data entry, and troubleshooting time.
- Program value, including adherence, clinical visibility, and reporting efficiency.
A practical buying decision should compare total cost of ownership over at least 6 to 12 months, not just the first purchase order. That is especially important when the device is intended to work with digital blood pressure monitors, wearable ECG monitors, or other connected home health tools.
What Buyers Should Evaluate Before Choosing a Smart Glucometer
Accuracy remains the first checkpoint. Smart features do not compensate for poor measurement consistency. Buyers should confirm whether the device meets relevant market requirements in its destination region and whether performance claims are supported by appropriate documentation. Quality teams should review sample handling instructions, strip storage conditions, operating temperature ranges, and calibration guidance because these affect real-world reliability just as much as software features.
The second checkpoint is connectivity quality. A smart glucometer is only useful if data transfer works consistently across patient environments. Procurement teams should ask about pairing time, sync reliability, offline storage capacity, and compatibility with major mobile operating systems. A device that stores 500 to 1,000 readings locally can be more resilient in low-connectivity settings than one that depends on immediate upload for every test.
The third checkpoint is user experience. Operators, clinicians, and end users may have different priorities. A patient may need a large screen and simple prompts, while a clinic may focus on dashboard reporting and data export. If onboarding takes more than 10 to 15 minutes per user, large-scale deployment becomes harder and support costs rise. Ease of use directly affects adherence, which in turn affects the business case.
Finally, enterprise buyers should evaluate service continuity. Ask about replacement timelines, firmware update processes, training materials, and distributor support capacity. In B2B healthcare procurement, service failure often costs more than hardware failure. A replacement cycle of 48 to 72 hours may be acceptable in some programs, but high-acuity monitoring workflows may require faster response.
Key selection criteria for procurement and quality teams
The matrix below helps compare candidates using operational and commercial criteria rather than product brochures alone.
For most B2B buyers, the best device is the one that balances 4 dimensions at once: accuracy, usability, interoperability, and serviceability. If one dimension is weak, the promised smart value can disappear quickly after rollout.
Questions to ask suppliers during evaluation
- How many readings can the device store if the app is not connected for 3 to 7 days?
- What is the typical onboarding time for first-time users?
- How are firmware updates delivered and documented?
- What replacement and after-sales support window is available for commercial buyers?
Implementation Risks, Common Missteps, and How to Avoid Them
Even well-chosen smart glucometers can underperform if deployment is poorly planned. One common mistake is treating the device as a plug-and-play upgrade without redesigning workflow. If staff still collect readings manually, or if patients are not shown how to sync data within the first 24 hours, the organization may pay for smart functionality it never uses. Technology value depends on process adoption.
Another risk is overbuying features. Some programs need only basic app logging and trend sharing, not full enterprise integration. Others require deeper compatibility with care management platforms. Buying beyond actual need raises cost, while underbuying creates future migration expense. A 2-stage procurement model often works well: pilot with 20 to 50 users, review data flow and adherence, then scale based on measured outcomes.
Quality and safety managers should also consider consumables and supply continuity. Test strips, lancets, batteries or charging accessories, and packaging consistency all affect field performance. A smart glucometer is not operationally smart if strip replenishment takes 3 weeks or if accessory compatibility changes without notice. Supply chain stability is part of device value, especially for distributors and regional healthcare networks.
Data governance is another practical issue. Buyers should clarify who owns the data, how long readings are stored, and what export formats are available. This matters when data must move between patient apps, provider systems, and reporting platforms. If data access is too limited, the device may create a digital silo rather than improving care coordination.
A practical 5-step rollout model
- Define the use case: home monitoring, outpatient follow-up, chronic care, or channel resale.
- Run a pilot for 2 to 6 weeks with measurable outcomes such as sync success and adherence rate.
- Train users with simple setup instructions, ideally under 10 minutes for first use.
- Review supply readiness for strips, replacement units, and technical support.
- Scale only after confirming workflow fit, patient usability, and data reporting quality.
Common misjudgments in buying decisions
- Assuming all Bluetooth-enabled devices deliver the same app experience.
- Ignoring strip logistics while focusing only on meter features.
- Approving hardware budget without planning training and support budget.
- Using a hospital workflow standard for home users with very different digital literacy levels.
A disciplined implementation plan reduces the chance that a premium device becomes a low-utilization asset. For financial approvers, this is often the deciding factor: not whether the meter is smart, but whether the organization is ready to use its smart capabilities effectively.
FAQ for Buyers, Distributors, and Healthcare Decision-Makers
Are smart glucometers suitable for every care setting?
No. They are most valuable where data sharing, trend review, and remote visibility influence care decisions. For simple in-clinic spot testing or cost-sensitive bulk supply projects, standard devices may still be the better fit. Smart models add the most value when readings need to be reviewed over 14-day, 30-day, or 90-day periods by more than one stakeholder.
What procurement metrics matter most besides price?
Buyers should track at least 5 metrics: onboarding time, sync success rate, reading storage capacity, consumables availability, and replacement service speed. These metrics often determine whether the device performs well after deployment. In many programs, a low-cost meter with poor adoption becomes more expensive than a premium device with smooth workflow integration.
How long does a typical commercial rollout take?
A pilot rollout can often begin within 2 to 4 weeks if inventory, user instructions, and software access are ready. A multi-site deployment may take 6 to 12 weeks depending on procurement cycles, user training, and system configuration. If the program includes additional devices such as blood pressure monitors or wearable ECG monitors, planning time may increase because interoperability checks are more complex.
Can smart glucometers improve adherence enough to justify the price?
They can, but only when reminders, dashboards, and caregiver visibility are actually used. Better adherence is not guaranteed by connectivity alone. The strongest results usually come from combining device simplicity, clear onboarding, and follow-up protocols. Buyers should evaluate adherence during a pilot rather than assuming the app itself will change user behavior.
Smart glucometers are worth the extra cost when they solve a real workflow problem, improve data visibility, and support measurable patient management outcomes. They are less compelling when bought purely for feature comparison without a defined care pathway or service model. For healthcare teams, distributors, and enterprise buyers building connected care programs, the smartest purchase is the one aligned with total cost of ownership, deployment readiness, and long-term monitoring goals. To explore sourcing options, integration priorities, or a tailored healthcare device strategy, contact us to discuss your application needs and get a more precise procurement roadmap.
Get weekly intelligence in your inbox.
No noise. No sponsored content. Pure intelligence.



























